QUESTION IMAGE
Question
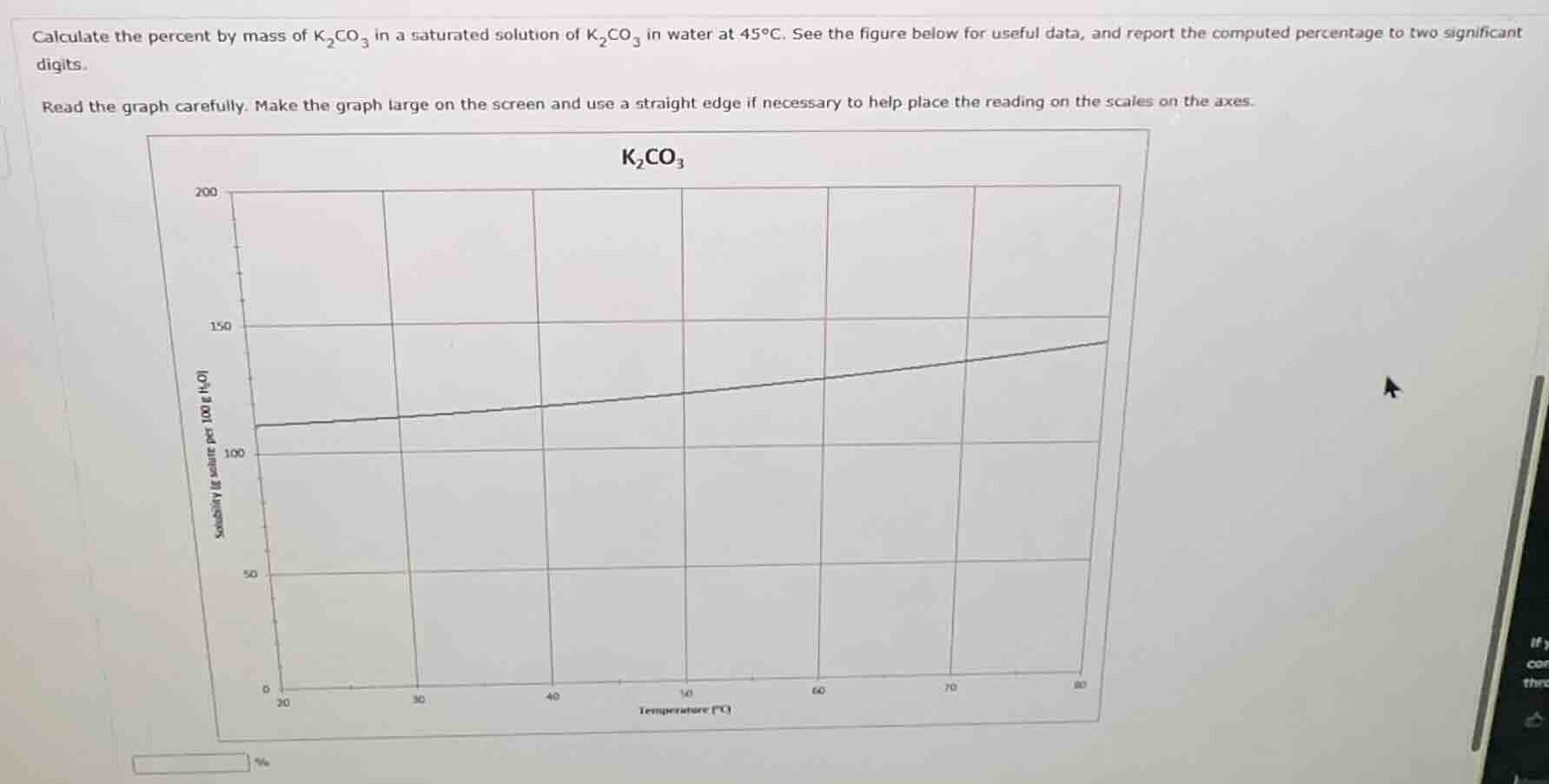
calculate the percent by mass of $\ce{k_{2}co_{3}}$ in a saturated solution of $\ce{k_{2}co_{3}}$ in water at $45{\hspace{0pt}}^{\circ }c$. see the figure below for useful data, and report the computed percentage to two significant digits.
read the graph carefully. make the graph large on the screen and use a straight edge if necessary to help place the reading on the scales on the axes.
Step1: Find solubility at 45°C
From the graph, at 45°C, the solubility of \(K_2CO_3\) (mass of solute per 100 g of water) is approximately 130 g (reading the graph: x - axis temperature, y - axis solubility g per 100 g \(H_2O\); at 45°C, y - value is ~130 g).
Step2: Calculate total mass of solution
Mass of solute (\(K_2CO_3\)) = 130 g, mass of solvent (water) = 100 g.
Total mass of solution = mass of solute + mass of solvent = \(130 + 100 = 230\) g.
Step3: Calculate percent by mass
Percent by mass formula: \(\text{Percent by mass} = \frac{\text{mass of solute}}{\text{mass of solution}} \times 100\%\)
Substitute values: \(\frac{130}{230} \times 100\% \approx 56.52\%\). Rounding to two significant digits: ~57% (or checking the graph reading more accurately: if solubility is ~135 g, then \(\frac{135}{235} \times 100 \approx 57.45\%\), still ~57% with two sig figs. Wait, maybe the graph at 45°C is ~130 - 135 g. Let's re - check: the y - axis at 45°C (between 40 and 50°C), the line is around 130 - 135. Let's take 130 g: \(\frac{130}{230} \approx 0.565\), times 100 is 56.5%, two sig figs is 57%? Wait, no: 130 has two sig figs? Wait, the graph's y - axis: the grid lines. Let's see, the y - axis has 0,50,100,150. At 45°C, the solubility is approximately 130 g (since between 100 and 150, closer to 130). So mass of solute = 130 g (two sig figs? Wait, 130 could be two or three, but when we calculate, \(\frac{130}{230}\): 130/230 = 0.565, times 100 is 56.5, two sig figs is 57%? Wait, no, maybe the correct reading is 130 g (as per graph). Alternatively, if the solubility is 130 g (solute) in 100 g water, then percent by mass is (130 / (130 + 100)) 100 = (130 / 230) 100 ≈ 56.5%, which rounds to 57% (two significant digits). Wait, but maybe the graph gives 130 g, so the calculation is as above.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\boxed{57\%}\) (or depending on graph reading precision, could be ~56% - 57%, but with two sig figs, 57% is appropriate)