QUESTION IMAGE
Question
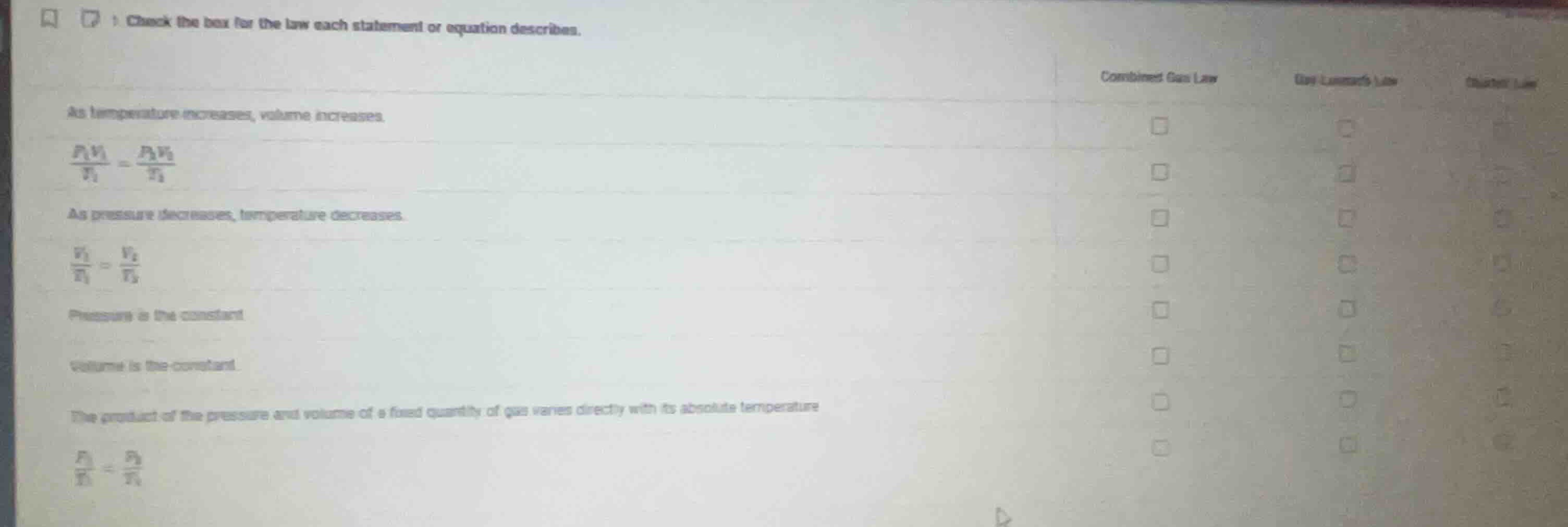
check the box for the law each statement or equation describes.
combined gas law
gay-lussacs law
charles law
as temperature increases, volume increases.
$\frac{p_1v_1}{t_1} = \frac{p_2v_2}{t_2}$
as pressure decreases, temperature decreases.
$\frac{v_1}{t_1} = \frac{v_2}{t_2}$
pressure is the constant.
volume is the constant.
the product of the pressure and volume of a fixed quantity of gas varies directly with its absolute temperature
$\frac{p_1}{t_1} = \frac{p_2}{t_2}$
Brief Explanations
- As temperature increases, volume increases: This describes Charles's Law, which states volume and absolute temperature are directly proportional at constant pressure.
- $\frac{P_1V_1}{T_1} = \frac{P_2V_2}{T_2}$: This is the mathematical form of the Combined Gas Law, which combines Boyle's, Charles's, and Gay-Lussac's laws.
- As pressure decreases, temperature decreases: This describes Gay-Lussac's Law, which states pressure and absolute temperature are directly proportional at constant volume.
- $\frac{V_1}{T_1} = \frac{V_2}{T_2}$: This is the mathematical form of Charles's Law, showing the direct relationship between volume and temperature at constant pressure.
- Pressure is the constant: This is the condition for Charles's Law, which only applies when pressure is held fixed.
- Volume is the constant: This is the condition for Gay-Lussac's Law, which only applies when volume is held fixed.
- The product of the pressure and volume of a fixed quantity of gas varies directly with its absolute temperature: This is the definition of the Combined Gas Law, which relates $P$, $V$, and $T$ for a fixed amount of gas.
- $\frac{P_1}{T_1} = \frac{P_2}{T_2}$: This is the mathematical form of Gay-Lussac's Law, showing the direct relationship between pressure and temperature at constant volume.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Statement/Equation | Combined Gas Law | Gay-Lussac's Law | Charles's Law |
|---|---|---|---|
| $\frac{P_1V_1}{T_1} = \frac{P_2V_2}{T_2}$ | $\boxtimes$ | $\square$ | $\square$ |
| As pressure decreases, temperature decreases. | $\square$ | $\boxtimes$ | $\square$ |
| $\frac{V_1}{T_1} = \frac{V_2}{T_2}$ | $\square$ | $\square$ | $\boxtimes$ |
| Pressure is the constant. | $\square$ | $\square$ | $\boxtimes$ |
| Volume is the constant. | $\square$ | $\boxtimes$ | $\square$ |
| The product of the pressure and volume of a fixed quantity of gas varies directly with its absolute temperature | $\boxtimes$ | $\square$ | $\square$ |
| $\frac{P_1}{T_1} = \frac{P_2}{T_2}$ | $\square$ | $\boxtimes$ | $\square$ |