QUESTION IMAGE
Question
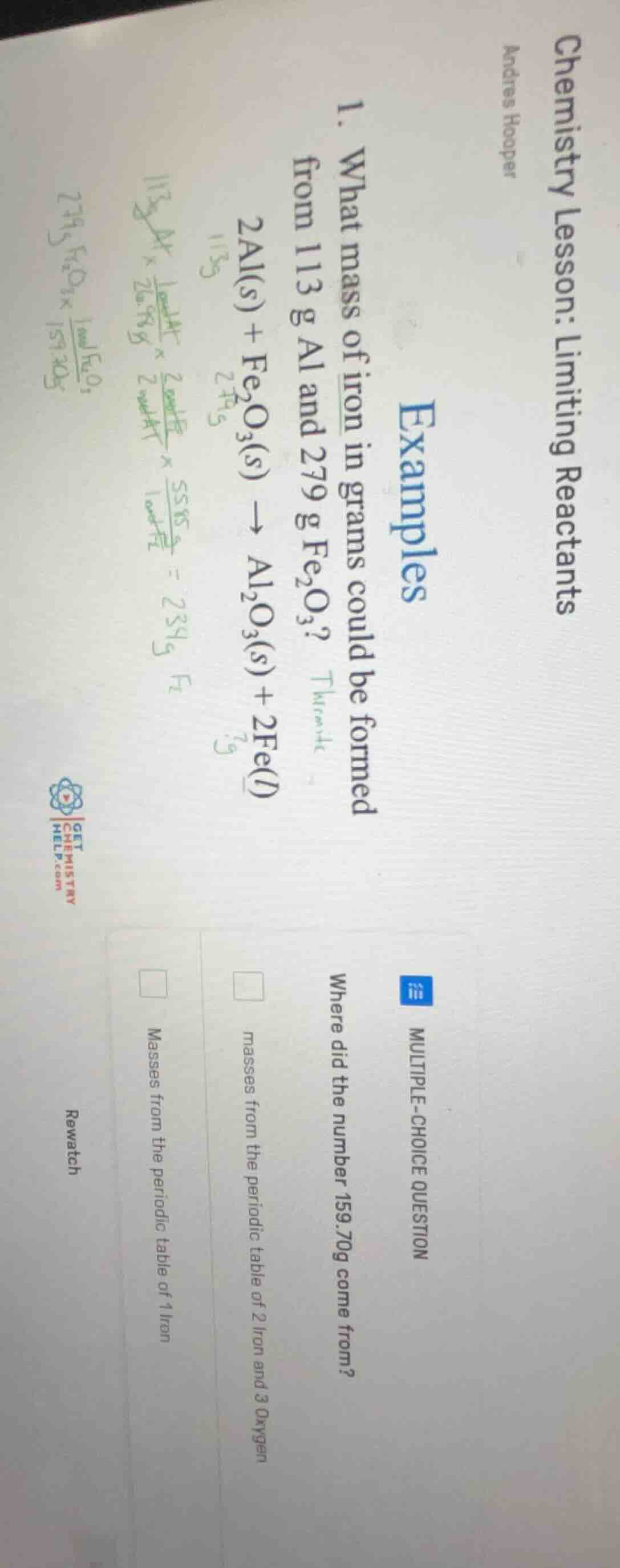
chemistry lesson: limiting reactants
andres hooper
examples
- what mass of iron in grams could be formed from 113 g al and 279 g $ce{fe_{2}o_{3}}$? thermite
$ce{2al(s) + fe_{2}o_{3}(s) -> al_{2}o_{3}(s) + 2fe(l)}$
multiple-choice question
where did the number 159.70g come from?
$square$ masses from the periodic table of 2 iron and 3 oxygen
$square$ masses from the periodic table of 1 iron
rewatch
Step1: Identify limiting reactant (Al)
Calculate Fe from 113 g Al:
$$113\ \text{g Al} \times \frac{1\ \text{mol Al}}{26.98\ \text{g Al}} \times \frac{2\ \text{mol Fe}}{2\ \text{mol Al}} \times \frac{55.85\ \text{g Fe}}{1\ \text{mol Fe}} = 234\ \text{g Fe}$$
Step2: Identify limiting reactant (Fe₂O₃)
Calculate Fe from 279 g Fe₂O₃:
$$279\ \text{g Fe}_2\text{O}_3 \times \frac{1\ \text{mol Fe}_2\text{O}_3}{159.70\ \text{g Fe}_2\text{O}_3} \times \frac{2\ \text{mol Fe}}{1\ \text{mol Fe}_2\text{O}_3} \times \frac{55.85\ \text{g Fe}}{1\ \text{mol Fe}} = 195\ \text{g Fe}$$
Step3: Determine smaller yield (limiting reactant)
The smaller mass of Fe is the maximum possible yield.
The value 159.70 g is the molar mass of iron(III) oxide ($\text{Fe}_2\text{O}_3$), calculated by summing the atomic masses of 2 iron atoms ($2 \times 55.85\ \text{g/mol}$) and 3 oxygen atoms ($3 \times 16.00\ \text{g/mol}$) from the periodic table.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
195 g of iron can be formed
---