QUESTION IMAGE
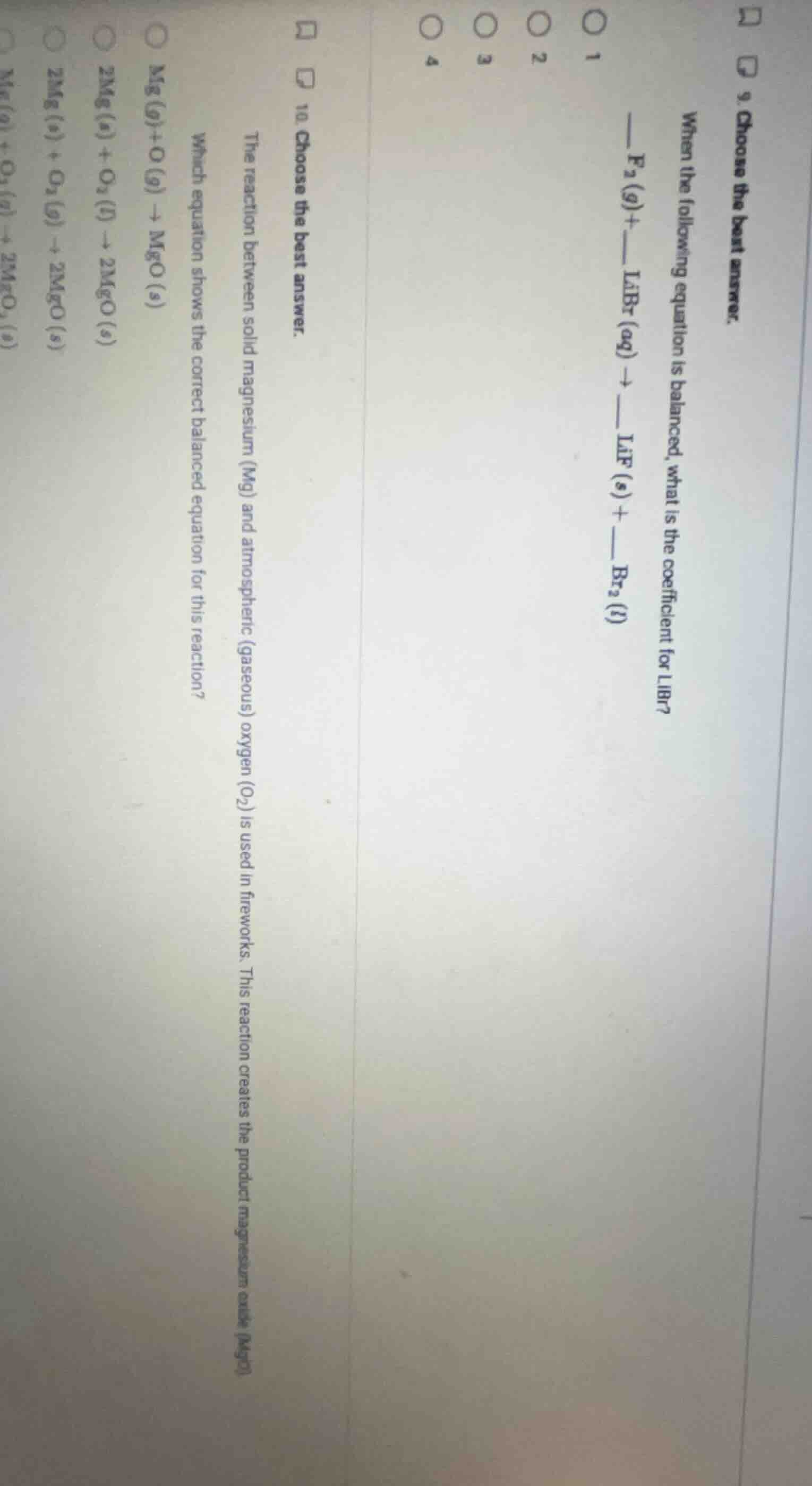
Question
- choose the best answer.
when the following equation is balanced, what is the coefficient for libr?
—— f₂(g) + —— libr(aq) → —— lif(s) + —— br₂(l)
1
2
3
4
- choose the best answer.
the reaction between solid magnesium (mg) and atmospheric (gaseous) oxygen (o₂) is used in fireworks. this reaction creates the product magnesium oxide (mgo).
which equation shows the correct balanced equation for this reaction?
mg(g) + o(g) → mgo(s)
2mg(g) + o₂(l) → 2mgo(s)
2mg(g) + o₂(g) → 2mgo(s)
mg(g) + o₂(g) → 2mgo₂(s)
Step1: Balance Br atoms
Count Br on right: 2 (in $\text{Br}_2$). So $\text{LiBr}$ needs coefficient 2 to get 2 Br on left.
Step2: Balance Li atoms
With $\text{LiBr}$ coefficient 2, Li on left is 2. So $\text{LiF}$ needs coefficient 2 to match Li on right.
Step3: Balance F atoms
$\text{LiF}$ has coefficient 2, so F on right is 2. $\text{F}_2$ has 2 F, so its coefficient is 1. Balanced equation:
---
Step1: Identify reactant states
Mg is solid ($s$), atmospheric $\text{O}_2$ is gas ($g$).
Step2: Balance Mg and O atoms
Unbalanced: $\text{Mg}(s) + \text{O}_2(g)
ightarrow \text{MgO}(s)$. Add 2 to Mg and MgO:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 2
- $2\text{Mg}(s) + \text{O}_2(g)
ightarrow 2\text{MgO}(s)$