QUESTION IMAGE
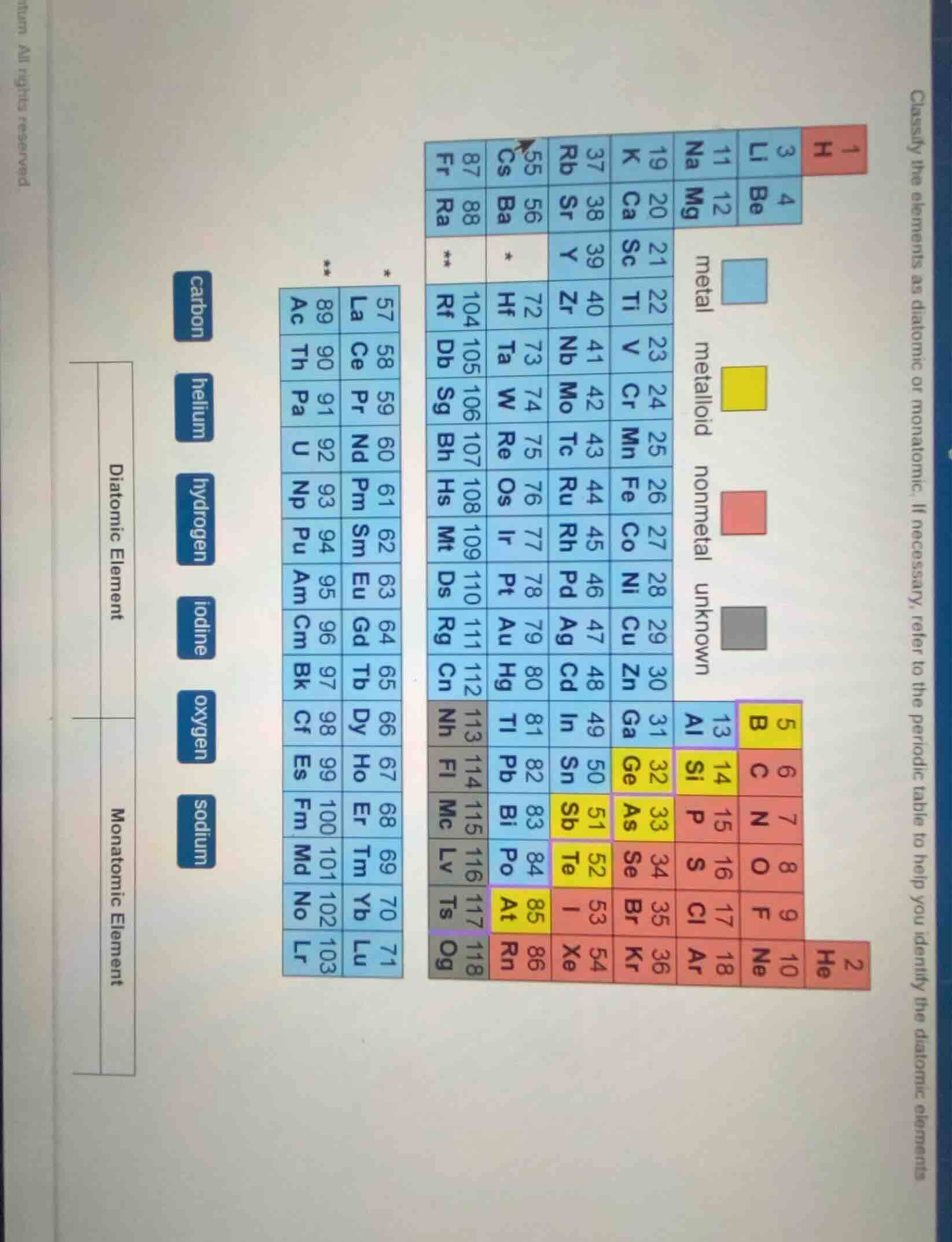
Question
classify the elements as diatomic or monatomic. if necessary, refer to the periodic table to help you identify the diatomic elements.
diatomic element
monatomic element
carbon
helium
hydrogen
iodine
oxygen
sodium
To solve this, we analyze each element:
Diatomic Elements (exist as \( X_2 \) molecules):
- Hydrogen (\( \text{H}_2 \)): Diatomic.
- Oxygen (\( \text{O}_2 \)): Diatomic.
- Iodine (\( \text{I}_2 \)): Diatomic (solid diatomic).
Monatomic Elements (exist as single atoms, mostly noble gases or metals):
- Helium (\( \text{He} \)): Noble gas, monatomic.
- Carbon (\( \text{C} \)): Monatomic (exists as atoms in structures like graphite/diamond).
- Sodium (\( \text{Na} \)): Metal, monatomic (exists as \( \text{Na}^+ \) in metallic lattice, but as single atoms in gas phase; here classified as monatomic for simplicity).
Final Classification:
| Diatomic Element | Monatomic Element |
|---|---|
| Oxygen | Carbon |
| Iodine | Sodium |
(Note: For elements like carbon, in standard definitions, it is polyatomic in bulk (e.g., \( \text{C}_60 \), graphite), but for basic classification, it is often grouped as monatomic here. Diatomic elements follow the \( \text{H}_2, \text{O}_2, \text{I}_2 \) pattern.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each element:
Diatomic Elements (exist as \( X_2 \) molecules):
- Hydrogen (\( \text{H}_2 \)): Diatomic.
- Oxygen (\( \text{O}_2 \)): Diatomic.
- Iodine (\( \text{I}_2 \)): Diatomic (solid diatomic).
Monatomic Elements (exist as single atoms, mostly noble gases or metals):
- Helium (\( \text{He} \)): Noble gas, monatomic.
- Carbon (\( \text{C} \)): Monatomic (exists as atoms in structures like graphite/diamond).
- Sodium (\( \text{Na} \)): Metal, monatomic (exists as \( \text{Na}^+ \) in metallic lattice, but as single atoms in gas phase; here classified as monatomic for simplicity).
Final Classification:
| Diatomic Element | Monatomic Element |
|---|---|
| Oxygen | Carbon |
| Iodine | Sodium |
(Note: For elements like carbon, in standard definitions, it is polyatomic in bulk (e.g., \( \text{C}_60 \), graphite), but for basic classification, it is often grouped as monatomic here. Diatomic elements follow the \( \text{H}_2, \text{O}_2, \text{I}_2 \) pattern.)