QUESTION IMAGE
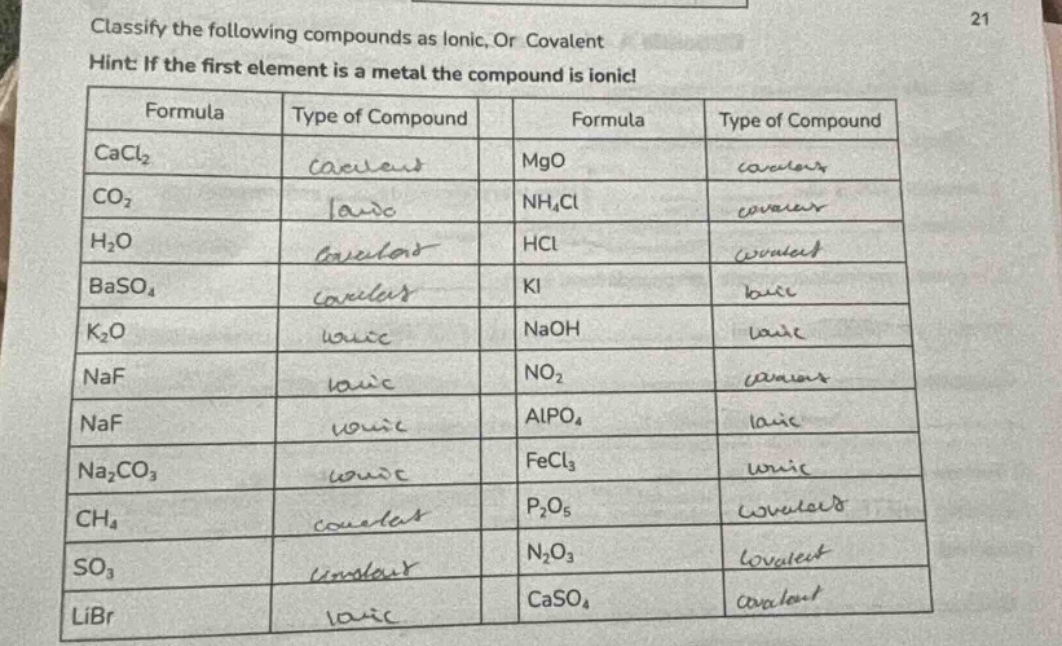
Question
classify the following compounds as ionic, or covalent
hint: if the first element is a metal the compound is ionic!
| formula | type of compound | formula | type of compound |
|---|---|---|---|
| co₂ | nh₄cl | ||
| h₂o | hcl | ||
| baso₄ | ki | ||
| k₂o | naoh | ||
| naf | no₂ | ||
| naf | alpo₄ | ||
| na₂co₃ | fecl₃ | ||
| ch₄ | p₂o₅ | ||
| so₃ | n₂o₃ | ||
| libr | caso₄ |
Step1: Identify ionic/covalent via metal rule
Ionic: Contains metal (or $NH_4^+$); Covalent: Nonmetals only.
Step2: Classify each compound one by one
- $CaCl_2$: Ca (metal) → Ionic
- $CO_2$: C, O (nonmetals) → Covalent
- $H_2O$: H, O (nonmetals) → Covalent
- $BaSO_4$: Ba (metal) → Ionic
- $K_2O$: K (metal) → Ionic
- $NaF$: Na (metal) → Ionic
- $NaF$: Na (metal) → Ionic
- $Na_2CO_3$: Na (metal) → Ionic
- $CH_4$: C, H (nonmetals) → Covalent
- $SO_3$: S, O (nonmetals) → Covalent
- $LiBr$: Li (metal) → Ionic
- $MgO$: Mg (metal) → Ionic
- $NH_4Cl$: $NH_4^+$ (ionic cation) → Ionic
- $HCl$: H, Cl (nonmetals) → Covalent
- $KI$: K (metal) → Ionic
- $NaOH$: Na (metal) → Ionic
- $NO_2$: N, O (nonmetals) → Covalent
- $AlPO_4$: Al (metal) → Ionic
- $FeCl_3$: Fe (metal) → Ionic
- $P_2O_5$: P, O (nonmetals) → Covalent
- $N_2O_3$: N, O (nonmetals) → Covalent
- $CaSO_4$: Ca (metal) → Ionic
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Formula | Type of Compound | Formula | Type of Compound |
|---|---|---|---|
| $CO_2$ | Covalent | $NH_4Cl$ | Ionic |
| $H_2O$ | Covalent | $HCl$ | Covalent |
| $BaSO_4$ | Ionic | $KI$ | Ionic |
| $K_2O$ | Ionic | $NaOH$ | Ionic |
| $NaF$ | Ionic | $NO_2$ | Covalent |
| $NaF$ | Ionic | $AlPO_4$ | Ionic |
| $Na_2CO_3$ | Ionic | $FeCl_3$ | Ionic |
| $CH_4$ | Covalent | $P_2O_5$ | Covalent |
| $SO_3$ | Covalent | $N_2O_3$ | Covalent |
| $LiBr$ | Ionic | $CaSO_4$ | Ionic |