QUESTION IMAGE
Question
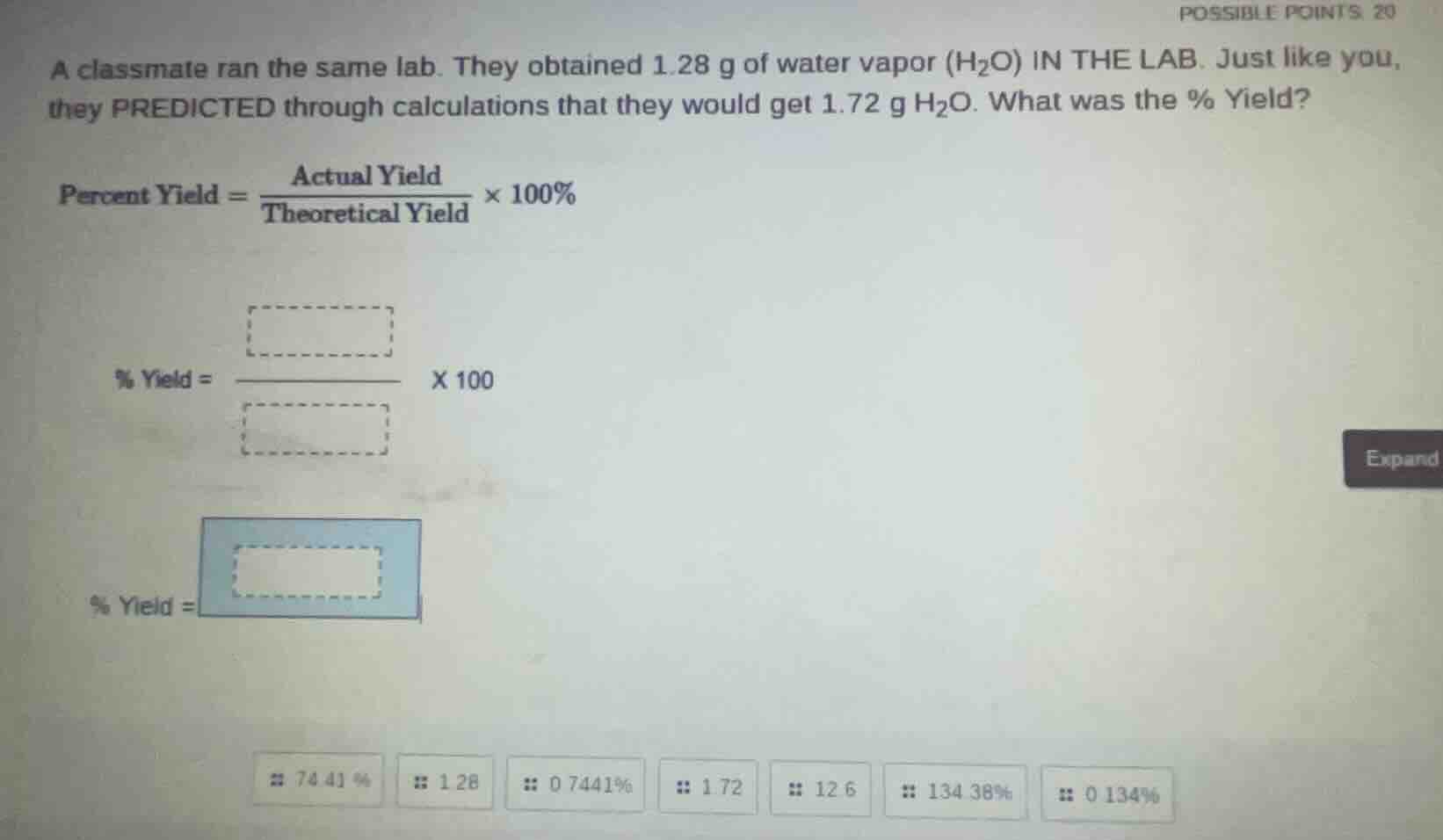
a classmate ran the same lab. they obtained 1.28 g of water vapor (h₂o) in the lab. just like you, they predicted through calculations that they would get 1.72 g h₂o. what was the % yield? percent yield = (actual yield / theoretical yield) × 100% % yield = (□ / □) × 100 % yield = □ 74.41 % 1.28 0.7441% 1.72 12.6 134.38% 0.134%
Step1: Identify Actual and Theoretical Yields
Actual Yield = 1.28 g (obtained in lab), Theoretical Yield = 1.72 g (predicted).
Step2: Plug into Percent Yield Formula
Percent Yield = $\frac{1.28}{1.72} \times 100\%$
Calculate $\frac{1.28}{1.72} \approx 0.7441$, then multiply by 100%: $0.7441 \times 100\% = 74.41\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
74.41%