QUESTION IMAGE
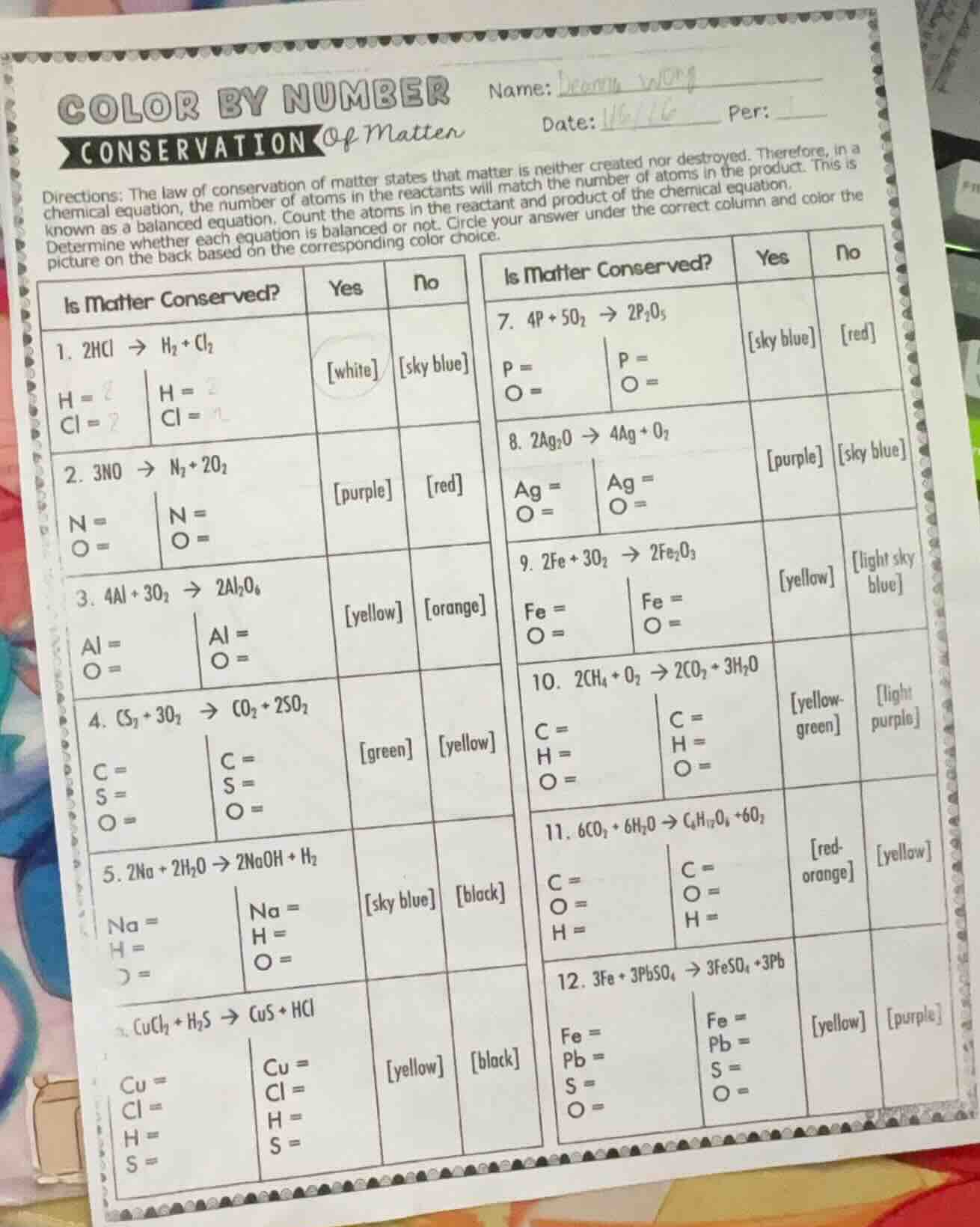
Question
color by number
conservation of matter
directions: the law of conservation of matter states that matter is neither created nor destroyed. therefore, in a chemical equation, the number of atoms in the reactants will match the number of atoms in the product. this is known as a balanced equation. count the atoms in the reactant and product of the chemical equation. determine whether each equation is balanced or not. circle your answer under the correct column and color the picture on the back based on the corresponding color choice.
- $2\ce{hcl} \
ightarrow \ce{h2} + \ce{cl2}$
reactant: $\ce{h}=2$, $\ce{cl}=2$; product: $\ce{h}=2$, $\ce{cl}=2$
is matter conserved? yes white no sky blue
- $3\ce{no} \
ightarrow \ce{n2} + 2\ce{o2}$
reactant: $\ce{n}=3$, $\ce{o}=3$; product: $\ce{n}=2$, $\ce{o}=4$
is matter conserved? yes purple no red
- $4\ce{al} + 3\ce{o2} \
ightarrow 2\ce{al2o6}$ (note: likely a typo, should be $\ce{al2o3}$)
reactant: $\ce{al}=4$, $\ce{o}=6$; product: $\ce{al}=4$, $\ce{o}=12$ (if $\ce{al2o6}$) or $\ce{o}=6$ (if $\ce{al2o3}$)
is matter conserved? yes yellow no orange
- $\ce{cs2} + 3\ce{o2} \
ightarrow \ce{co2} + 2\ce{so2}$
reactant: $\ce{c}=1$, $\ce{s}=2$, $\ce{o}=6$; product: $\ce{c}=1$, $\ce{s}=2$, $\ce{o}=6$
is matter conserved? yes green no yellow
- $2\ce{na} + 2\ce{h2o} \
ightarrow 2\ce{naoh} + \ce{h2}$
reactant: $\ce{na}=2$, $\ce{h}=4$, $\ce{o}=2$; product: $\ce{na}=2$, $\ce{h}=4$, $\ce{o}=2$
is matter conserved? yes sky blue no black
- $\ce{cucl2} + \ce{h2s} \
ightarrow \ce{cus} + \ce{hcl}$ (unbalanced, should be $2\ce{hcl}$)
reactant: $\ce{cu}=1$, $\ce{cl}=2$, $\ce{h}=2$, $\ce{s}=1$; product: $\ce{cu}=1$, $\ce{cl}=1$, $\ce{h}=1$, $\ce{s}=1$
is matter conserved? yes yellow no black
- $4\ce{p} + 5\ce{o2} \
ightarrow 2\ce{p2o5}$
reactant: $\ce{p}=4$, $\ce{o}=10$; product: $\ce{p}=4$, $\ce{o}=10$
is matter conserved? yes sky blue no red
- $2\ce{ag2o} \
ightarrow 4\ce{ag} + \ce{o2}$
reactant: $\ce{ag}=4$, $\ce{o}=2$; product: $\ce{ag}=4$, $\ce{o}=2$
is matter conserved? yes purple no sky blue
- $2\ce{fe} + 3\ce{o2} \
ightarrow 2\ce{fe2o3}$ (unbalanced, should be $4\ce{fe} + 3\ce{o2} \
ightarrow 2\ce{fe2o3}$)
reactant: $\ce{fe}=2$, $\ce{o}=6$; product: $\ce{fe}=4$, $\ce{o}=6$
is matter conserved? yes yellow no light sky blue
- $2\ce{ch4} + \ce{o2} \
ightarrow 2\ce{co2} + 3\ce{h2o}$ (unbalanced, should be $2\ce{ch4} + 4\ce{o2} \
ightarrow 2\ce{co2} + 4\ce{h2o}$ or adjusted)
reactant: $\ce{c}=2$, $\ce{h}=8$, $\ce{o}=2$; product: $\ce{c}=2$, $\ce{h}=6$, $\ce{o}=7$
is matter conserved? yes yellow - green no light purple
- $6\ce{co2} + 6\ce{h2o} \
ightarrow \ce{c6h12o6} + 6\ce{o2}$
reactant: $\ce{c}=6$, $\ce{o}=18$, $\ce{h}=12$; product: $\ce{c}=6$, $\ce{o}=12$, $\ce{h}=12$ (wait, $\ce{c6h12o6}$ has $\ce{o}=6$, plus $6\ce{o2}$ (12 o) → total o=18, same as reactant 62 + 61 = 18. correct balancing: $6\ce{co2} + 6\ce{h2o} \
ightarrow \ce{c6h12o6} + 6\ce{o2}$)
reactant: $\ce{c}=6$, $\ce{o}=18$, $\ce{h}=12$; product: $\ce{c}=6$, $\ce{o}=18$, $\ce{h}=12$
is matter conserved? yes red - orange no yellow
- $3\ce{fe} + 3\ce{pbso4} \
ightarrow 3\ce{feso4} + 3\ce{pb}$
reactant: $\ce{fe}=3$, $\ce{pb}=3$, $\ce{s}=3$, $\ce{o}=12$; product: $\ce{fe}=3$, $\ce{pb}=3$, $\ce{s}=3$, $\ce{o}=12$
is matter conserved? yes yellow no purple
Step1: Count reactant/product atoms (1)
Reactants: $H=2, Cl=2$; Products: $H=2, Cl=2$
Step2: Balance check (1)
Atoms equal → Balanced (Yes, white)
Step3: Count reactant/product atoms (2)
Reactants: $N=3, O=3$; Products: $N=2, O=4$
Step4: Balance check (2)
Atoms unequal → Unbalanced (No, red)
Step5: Count reactant/product atoms (3)
Reactants: $Al=4, O=6$; Products: $Al=4, O=24$
Step6: Balance check (3)
Atoms unequal → Unbalanced (No, orange)
Step7: Count reactant/product atoms (4)
Reactants: $C=1, S=2, O=6$; Products: $C=1, S=2, O=6$
Step8: Balance check (4)
Atoms equal → Balanced (Yes, green)
Step9: Count reactant/product atoms (5)
Reactants: $Na=2, H=4, O=2$; Products: $Na=2, H=4, O=2$
Step10: Balance check (5)
Atoms equal → Balanced (Yes, sky blue)
Step11: Count reactant/product atoms (6)
Reactants: $Cu=1, Cl=2, H=2, S=1$; Products: $Cu=1, Cl=1, H=2, S=1$
Step12: Balance check (6)
Atoms unequal → Unbalanced (No, black)
Step13: Count reactant/product atoms (7)
Reactants: $P=4, O=10$; Products: $P=4, O=10$
Step14: Balance check (7)
Atoms equal → Balanced (Yes, sky blue)
Step15: Count reactant/product atoms (8)
Reactants: $Ag=4, O=2$; Products: $Ag=4, O=2$
Step16: Balance check (8)
Atoms equal → Balanced (Yes, purple)
Step17: Count reactant/product atoms (9)
Reactants: $Fe=2, O=6$; Products: $Fe=4, O=6$
Step18: Balance check (9)
Atoms unequal → Unbalanced (No, light sky blue)
Step19: Count reactant/product atoms (10)
Reactants: $C=2, H=8, O=2$; Products: $C=2, H=6, O=7$
Step20: Balance check (10)
Atoms unequal → Unbalanced (No, light purple)
Step21: Count reactant/product atoms (11)
Reactants: $C=6, O=18, H=12$; Products: $C=6, O=18, H=12$
Step22: Balance check (11)
Atoms equal → Balanced (Yes, red-orange)
Step23: Count reactant/product atoms (12)
Reactants: $Fe=3, Pb=3, S=3, O=12$; Products: $Fe=3, Pb=3, S=3, O=12$
Step24: Balance check (12)
Atoms equal → Balanced (Yes, yellow)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Yes (white)
- No (red)
- No (orange)
- Yes (green)
- Yes (sky blue)
- No (black)
- Yes (sky blue)
- Yes (purple)
- No (light sky blue)
- No (light purple)
- Yes (red-orange)
- Yes (yellow)
With atom counts for each equation:
- Reactants: $H=2, Cl=2$; Products: $H=2, Cl=2$
- Reactants: $N=3, O=3$; Products: $N=2, O=4$
- Reactants: $Al=4, O=6$; Products: $Al=4, O=24$
- Reactants: $C=1, S=2, O=6$; Products: $C=1, S=2, O=6$
- Reactants: $Na=2, H=4, O=2$; Products: $Na=2, H=4, O=2$
- Reactants: $Cu=1, Cl=2, H=2, S=1$; Products: $Cu=1, Cl=1, H=2, S=1$
- Reactants: $P=4, O=10$; Products: $P=4, O=10$
- Reactants: $Ag=4, O=2$; Products: $Ag=4, O=2$
- Reactants: $Fe=2, O=6$; Products: $Fe=4, O=6$
- Reactants: $C=2, H=8, O=2$; Products: $C=2, H=6, O=7$
- Reactants: $C=6, O=18, H=12$; Products: $C=6, O=18, H=12$
- Reactants: $Fe=3, Pb=3, S=3, O=12$; Products: $Fe=3, Pb=3, S=3, O=12$