QUESTION IMAGE
Question
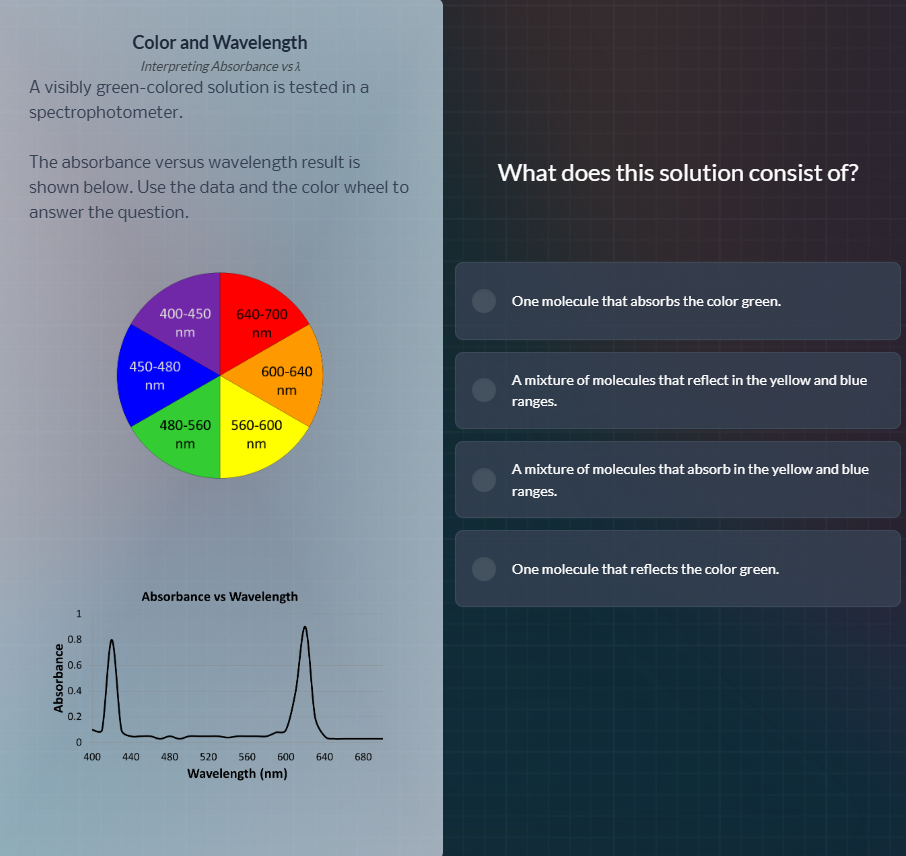
color and wavelengthinterpreting absorbance vs λa visibly green-colored solution is tested in a spectrophotometer.the absorbance versus wavelength result is shown below. use the data and the color wheel to answer the question.what does this solution consist of?one molecule that absorbs the color green.a mixture of molecules that reflect in the yellow and blue ranges.a mixture of molecules that absorb in the yellow and blue ranges.one molecule that reflects the color green.
- From the absorbance graph, the solution strongly absorbs wavelengths around 400-440 nm (blue range) and 620-660 nm (red/orange range, adjacent to yellow on the color wheel).
- Visually observed colors of solutions are the complementary (reflected/transmitted) colors of the wavelengths they absorb. A green solution reflects green light, meaning it absorbs the complementary colors: red/orange and blue (which correspond to the yellow-blue range's complementary absorbed wavelengths).
- The single absorbance curve with two peaks indicates one molecule is responsible for both absorption bands, not a mixture.
- The solution appears green because it reflects green light, absorbing the non-green complementary wavelengths.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
One molecule that reflects the color green.