QUESTION IMAGE
Question
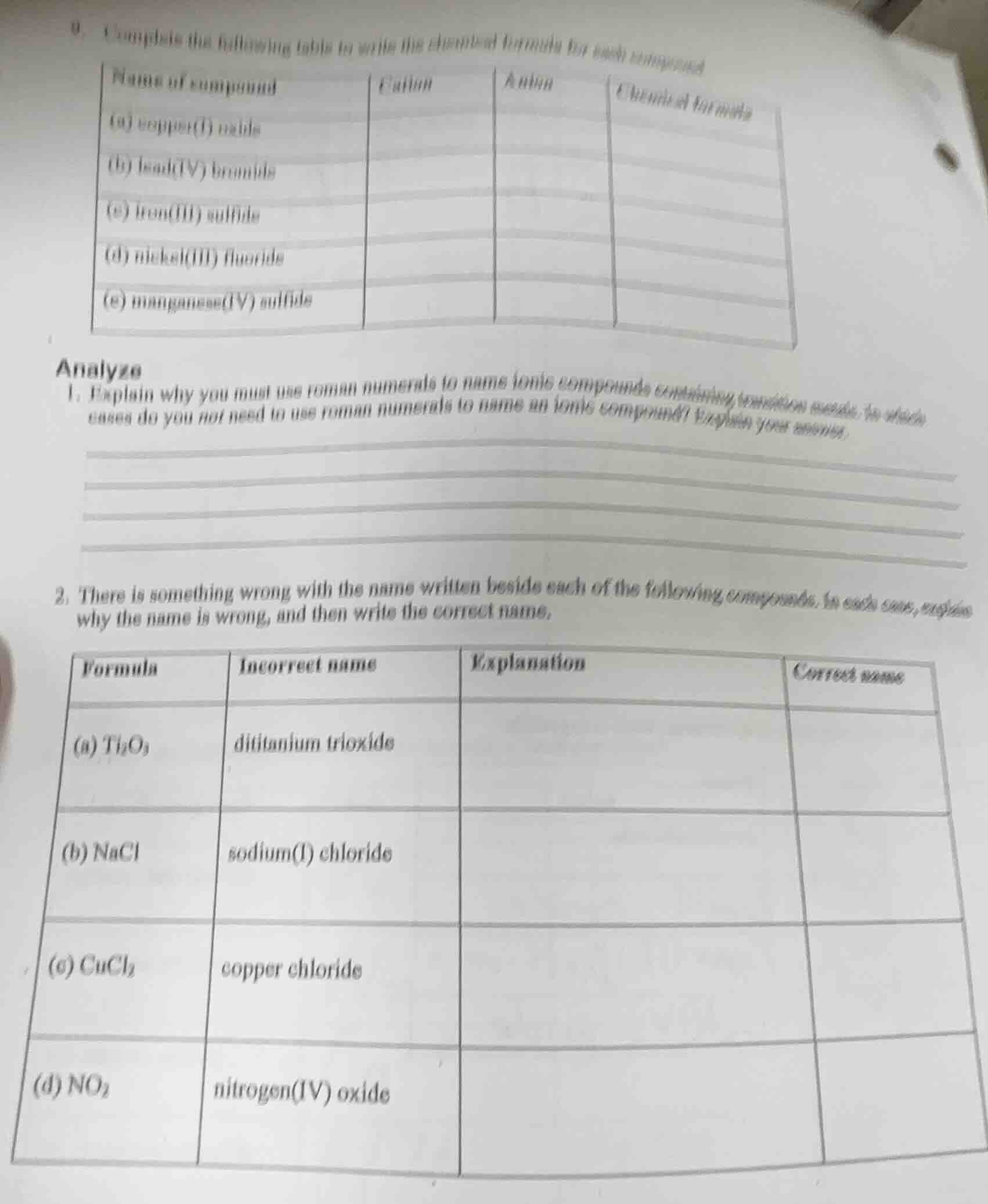
- complete the following table to write the chemical formula for each compound
| name of compound | cation | anion | chemical formula |
|---|---|---|---|
| (b) lead(iv) bromide | |||
| (c) iron(iii) sulfide | |||
| (d) nickel(iii) fluoride | |||
| (e) manganese(iv) sulfide |
analyze
- explain why you must use roman numerals to name ionic compounds containing transition metals. in which cases do you not need to use roman numerals to name an ionic compound? explain your answer.
- there is something wrong with the name written beside each of the following compounds. in each case, explain why the name is wrong, and then write the correct name.
| formula | incorrect name | explanation | correct name |
|---|---|---|---|
| (b) nacl | sodium(i) chloride | ||
| (c) cucl₂ | copper chloride | ||
| (d) no₂ | nitrogen(iv) oxide |
First Table: Complete the Chemical Formula Table
Step1: Identify cation/anion charges
For each compound, extract the cation (with charge from Roman numeral) and anion (fixed charge):
(a) Copper(I) oxide: $\text{Cu}^+$, $\text{O}^{2-}$
(b) Lead(IV) bromide: $\text{Pb}^{4+}$, $\text{Br}^-$
(c) Iron(III) sulfide: $\text{Fe}^{3+}$, $\text{S}^{2-}$
(d) Nickel(III) fluoride: $\text{Ni}^{3+}$, $\text{F}^-$
(e) Manganese(IV) sulfide: $\text{Mn}^{4+}$, $\text{S}^{2-}$
Step2: Balance charges for neutrality
Use crisscross method to get subscripts:
(a) $2\times(+1) + 1\times(-2) = 0$ → $\text{Cu}_2\text{O}$
(b) $1\times(+4) + 4\times(-1) = 0$ → $\text{PbBr}_4$
(c) $2\times(+3) + 3\times(-2) = 0$ → $\text{Fe}_2\text{S}_3$
(d) $1\times(+3) + 3\times(-1) = 0$ → $\text{NiF}_3$
(e) $1\times(+4) + 2\times(-2) = 0$ → $\text{MnS}_2$
Transition metals can form multiple cations with different charges; Roman numerals specify the cation's charge to avoid ambiguity (e.g., iron can be $\text{Fe}^{2+}$ or $\text{Fe}^{3+}$). Roman numerals are not needed for ionic compounds containing group 1/2 metals (alkali/alkaline earth metals) or aluminum, as these elements only form one fixed-charge cation (e.g., $\text{Na}^+$, $\text{Mg}^{2+}$).
Step1: Assess each formula/name pair
For each entry, identify naming rules violated:
(a) $\text{Ti}_2\text{O}_3$: Ti is a transition metal; use Roman numerals, not prefixes.
(b) $\text{NaCl}$: Na is group 1, fixed charge; no Roman numeral needed.
(c) $\text{CuCl}_2$: Cu is a transition metal; charge must be specified.
(d) $\text{NO}_2$: It is a covalent compound; use prefixes, not Roman numerals.
Step2: Calculate charges (ionic) or apply covalent rules
(a) Ti charge: $\frac{2\times(-3)}{2}=+3$ → titanium(III) oxide
(b) Na only forms $\text{Na}^+$ → sodium chloride
(c) Cu charge: $+2$ (from $2\times(-1)$) → copper(II) chloride
(d) Covalent: 1 N, 2 O → nitrogen dioxide
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Name of compound | Cation | Anion | Chemical Formula |
|---|---|---|---|
| (b) lead(IV) bromide | $\text{Pb}^{4+}$ | $\text{Br}^-$ | $\text{PbBr}_4$ |
| (c) iron(III) sulfide | $\text{Fe}^{3+}$ | $\text{S}^{2-}$ | $\text{Fe}_2\text{S}_3$ |
| (d) nickel(III) fluoride | $\text{Ni}^{3+}$ | $\text{F}^-$ | $\text{NiF}_3$ |
| (e) manganese(IV) sulfide | $\text{Mn}^{4+}$ | $\text{S}^{2-}$ | $\text{MnS}_2$ |
---