QUESTION IMAGE
Question
complete the sentences to explain the difference
match the items in the left column to the appropriate blanks in the sentences on the right.
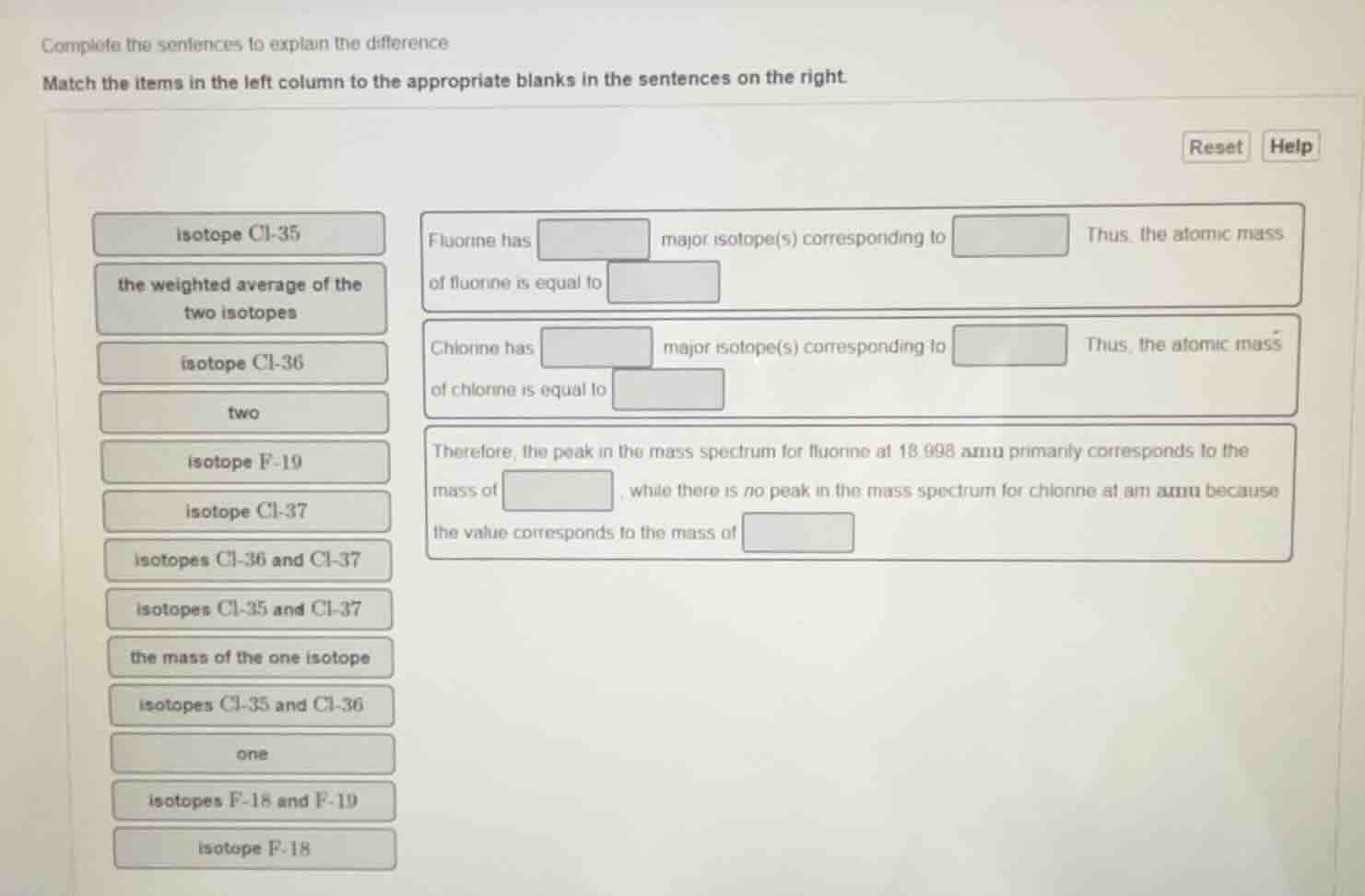
isotope cl-35
fluorine has major isotope(s) corresponding to thus, the atomic mass
the weighted average of the
of fluorine is equal to
two isotopes
chlorine has major isotope(s) corresponding to thus, the atomic mass
isotope cl-36
of chlorine is equal to
two
therefore, the peak in the mass spectrum for fluorine at 18.998 amu primarily corresponds to the
isotope f-19
mass of, while there is no peak in the mass spectrum for chlorine at am amu because
isotope cl-37
the value corresponds to the mass of
isotopes cl-36 and cl-37
isotopes cl-35 and cl-37
the mass of the one isotope
isotopes cl-35 and cl-36
one
isotopes f-18 and f-19
isotope f-18
- For fluorine: Fluorine has one major isotope (F - 19) as it is relatively stable with this single major isotope. So the number of major isotopes is "one", the isotope is "isotope F - 19", and the atomic mass is "the mass of the one isotope" since there's only one major isotope contributing.
- For chlorine: Chlorine has two major isotopes (Cl - 35 and Cl - 37). So the number of major isotopes is "two", the isotopes are "isotopes Cl - 35 and Cl - 37", and the atomic mass is "the weighted average of the two isotopes" as it is the average of these two isotopes' masses considering their abundances.
- For the mass spectrum: The peak for fluorine at 18.998 amu corresponds to "isotope F - 19" as that's its major isotope. For chlorine, the mass corresponding to no peak (like Cl - 36 which is not a major isotope) would be "isotope Cl - 36" (assuming the "am amu" is a typo, maybe 36 amu for Cl - 36 which is not a major isotope, so no peak for it as major isotopes are 35 and 37).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Fluorine has $\boldsymbol{\text{one}}$ major isotope(s) corresponding to $\boldsymbol{\text{isotope F - 19}}$. Thus, the atomic mass of fluorine is equal to $\boldsymbol{\text{the mass of the one isotope}}$.
- Chlorine has $\boldsymbol{\text{two}}$ major isotope(s) corresponding to $\boldsymbol{\text{isotopes Cl - 35 and Cl - 37}}$. Thus, the atomic mass of chlorine is equal to $\boldsymbol{\text{the weighted average of the two isotopes}}$.
- Therefore, the peak in the mass spectrum for fluorine at 18.998 amu primarily corresponds to the mass of $\boldsymbol{\text{isotope F - 19}}$, while there is no peak in the mass spectrum for chlorine at (assuming 36 amu) amu because the value corresponds to the mass of $\boldsymbol{\text{isotope Cl - 36}}$.