QUESTION IMAGE
Question
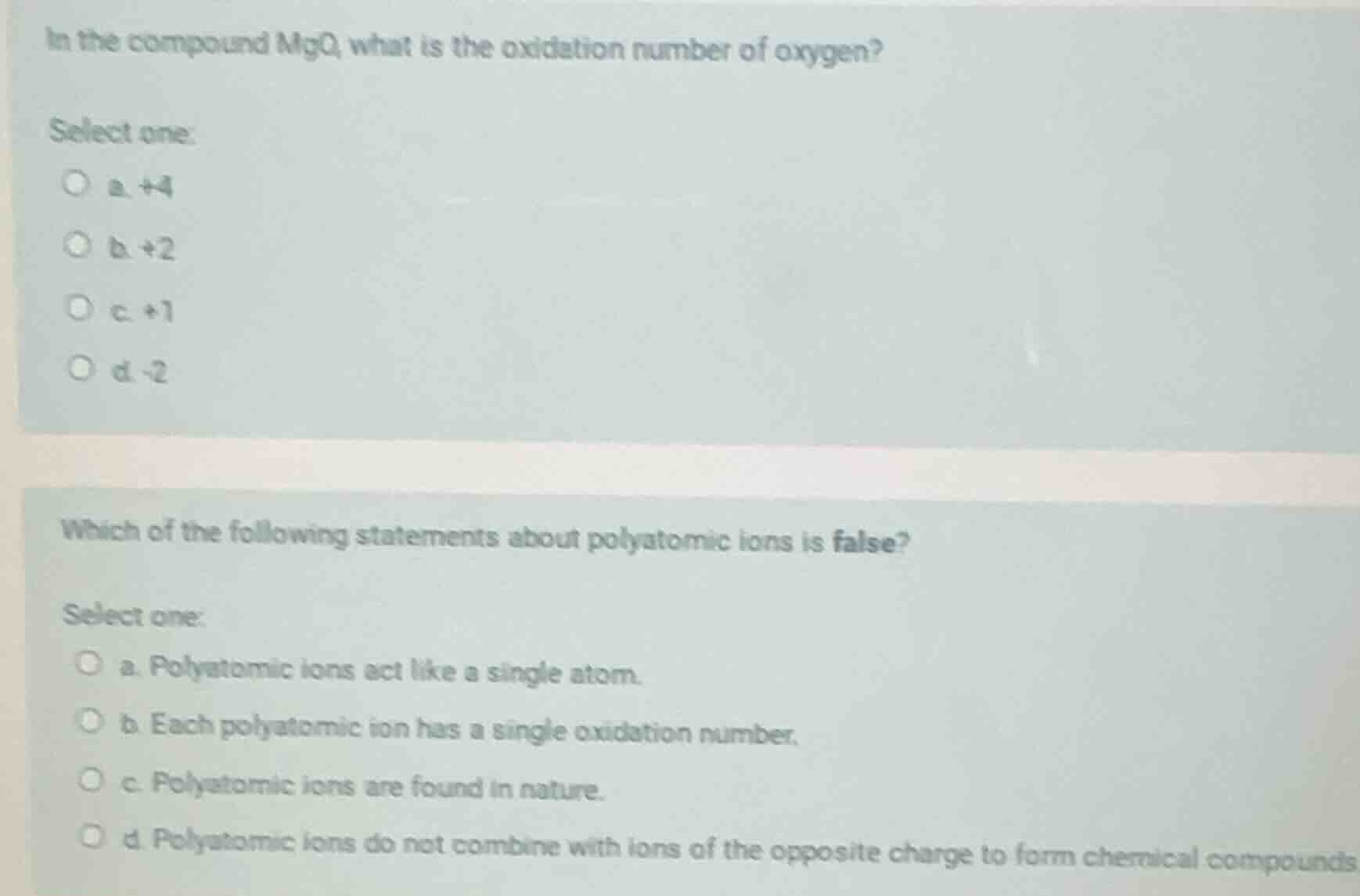
in the compound mgo, what is the oxidation number of oxygen?
select one:
a. +4
b. +2
c. +1
d. -2
which of the following statements about polyatomic ions is false?
select one:
a. polyatomic ions act like a single atom.
b. each polyatomic ion has a single oxidation number.
c. polyatomic ions are found in nature.
d. polyatomic ions do not combine with ions of the opposite charge to form chemical compounds
Step1: Solve first question
MgO is a neutral compound, so total oxidation number is 0. Mg has oxidation number +2. Let oxidation number of O be $x$.
$+2 + x = 0$
$x = 0 - 2 = -2$
Step2: Solve second question
Analyze each option:
- a: Polyatomic ions act as a single charged unit, similar to a single ion/atom in reactions. True.
- b: Each polyatomic ion has a fixed overall oxidation number. True.
- c: Polyatomic ions like carbonate exist in nature. True.
- d: Polyatomic ions combine with opposite charges to form compounds (e.g., $Na_2CO_3$). False.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- d. -2
- d. Polyatomic ions do not combine with ions of the opposite charge to form chemical compounds