QUESTION IMAGE
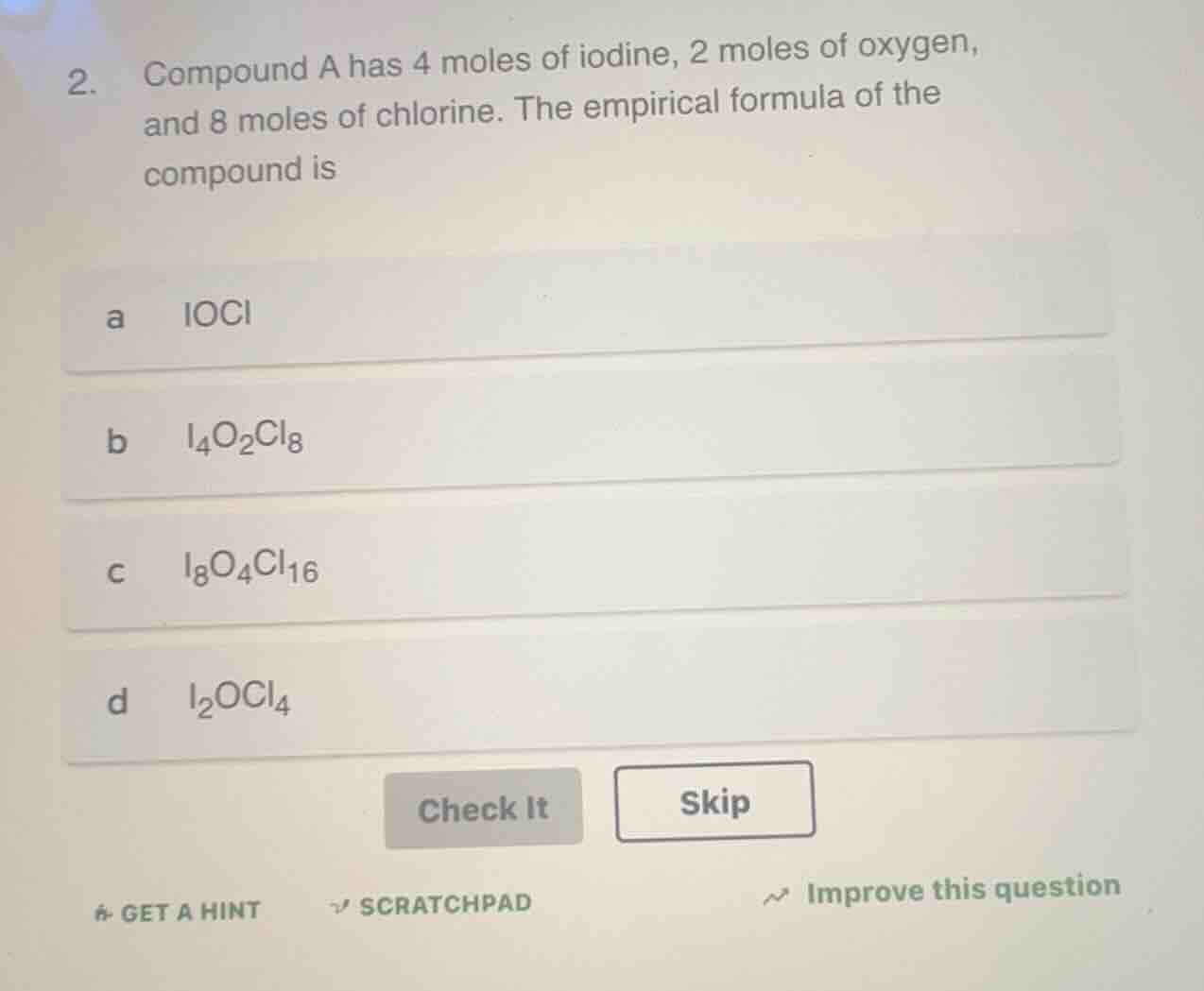
Question
- compound a has 4 moles of iodine, 2 moles of oxygen, and 8 moles of chlorine. the empirical formula of the compound is
a iocl
b i₄o₂cl₈
c i₈o₄cl₁₆
d i₂ocl₄
Step1: Find smallest mole value
The moles are: I=4, O=2, Cl=8. The smallest value is 2.
Step2: Divide all moles by smallest
For Iodine: $\frac{4}{2} = 2$
For Oxygen: $\frac{2}{2} = 1$
For Chlorine: $\frac{8}{2} = 4$
Step3: Write empirical formula
Use the resulting whole numbers as subscripts.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. $\text{I}_2\text{OCl}_4$