QUESTION IMAGE
Question
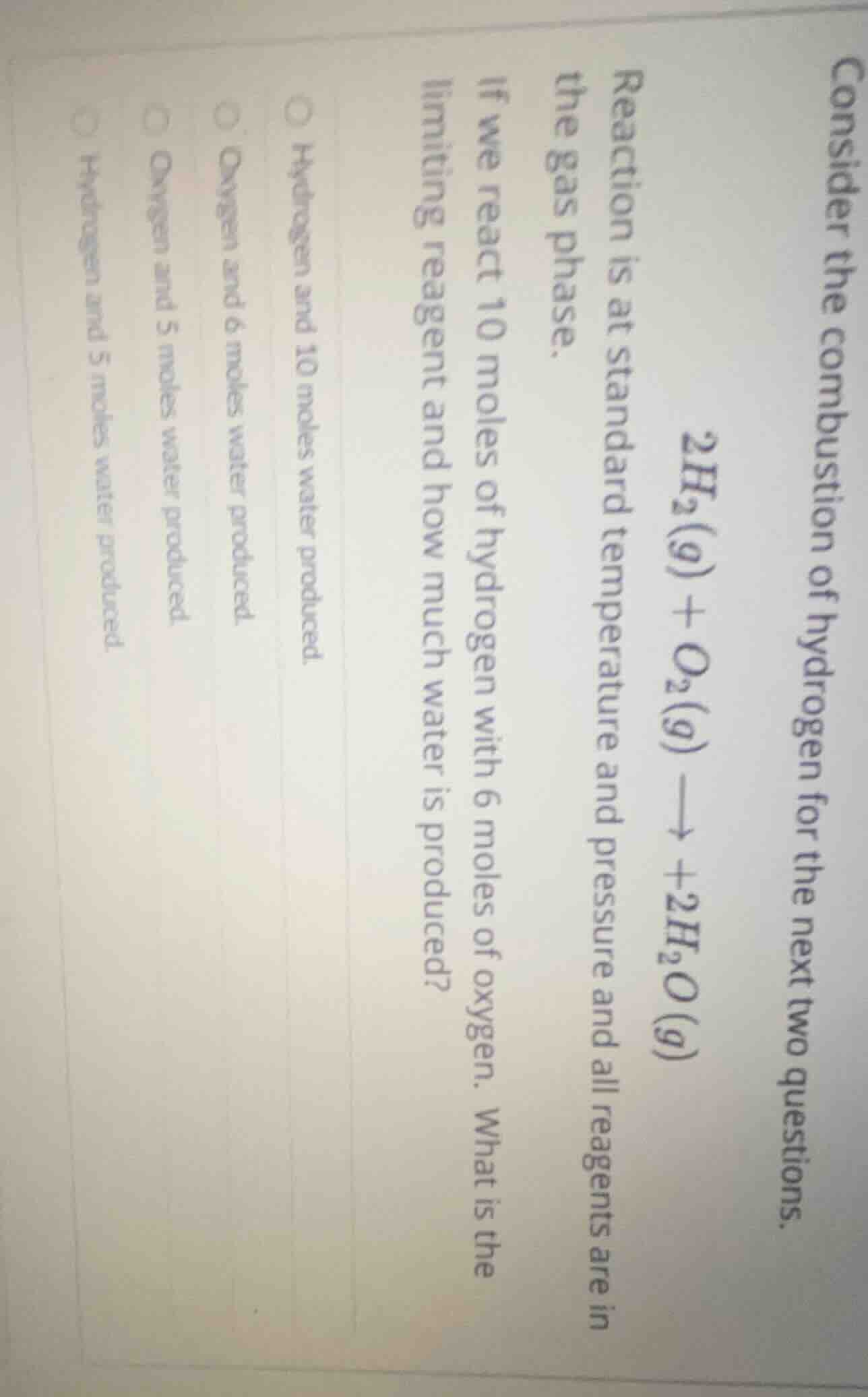
consider the combustion of hydrogen for the next two questions.
$2h_2(g) + o_2(g) \
ightarrow +2h_2o(g)$
reaction is at standard temperature and pressure and all reagents are in the gas phase.
if we react 10 moles of hydrogen with 6 moles of oxygen. what is the limiting reagent and how much water is produced?
\bigcirc hydrogen and 10 moles water produced.
\bigcirc oxygen and 6 moles water produced.
\bigcirc oxygen and 5 moles water produced.
\bigcirc hydrogen and 5 moles water produced.
Step1: Identify mole ratio from reaction
From $2H_2(g) + O_2(g)
ightarrow 2H_2O(g)$, the mole ratio of $H_2:O_2$ is $2:1$.
Step2: Calculate $O_2$ needed for 10 mol $H_2$
Moles of $O_2$ required = $\frac{10}{2} = 5$ mol
Step3: Compare required vs available $O_2$
Available $O_2$ = 6 mol, which is more than 5 mol. So $H_2$ is limiting.
Step4: Calculate moles of $H_2O$ produced
Mole ratio of $H_2:H_2O$ is $1:1$, so moles of $H_2O$ = 10 mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Hydrogen and 10 moles water produced.