QUESTION IMAGE
Question
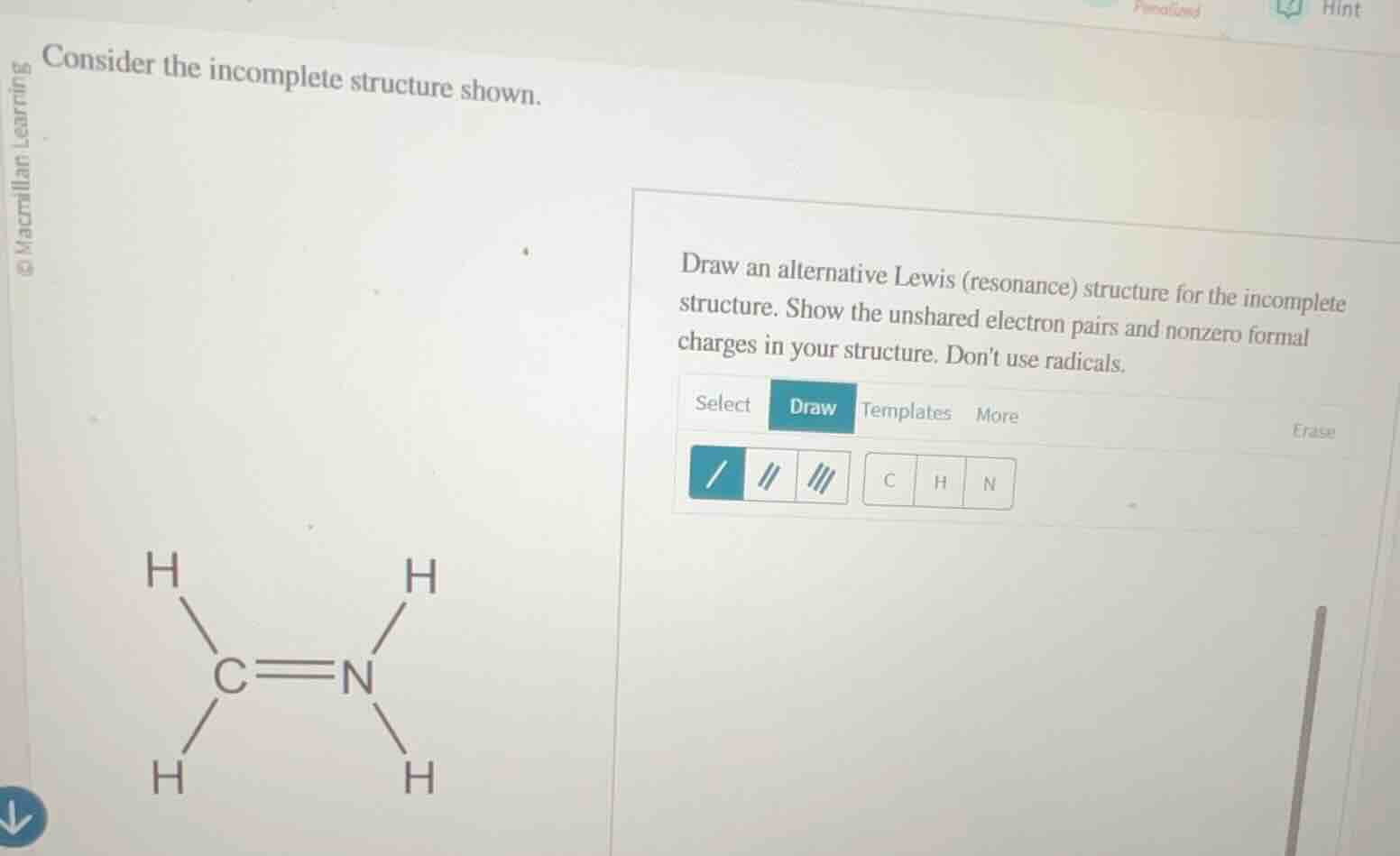
consider the incomplete structure shown. draw an alternative lewis (resonance) structure for the incomplete structure. show the unshared electron pairs and nonzero formal charges in your structure. dont use radicals. the given structure has c double bonded to n, with c bonded to two h atoms and n bonded to two h atoms.
Step1: Calculate formal charges
For original structure:
C: $4 - (3 + \frac{4}{2}) = -1$
N: $5 - (3 + \frac{4}{2}) = +1$
Step2: Rearrange bonding electrons
Move lone pair from C to form triple bond, add lone pair to N.
New structure: $H_2C^- \equiv N^+H_2$ (with N having 1 unshared pair, C having 0 unshared pairs)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The resonance structure is:
H
|
H - C ≡ N - H
|
HWith a -1 formal charge on the carbon atom and a +1 formal charge on the nitrogen atom; the nitrogen atom has one unshared electron pair, and the carbon atom has no unshared electron pairs.