QUESTION IMAGE
Question
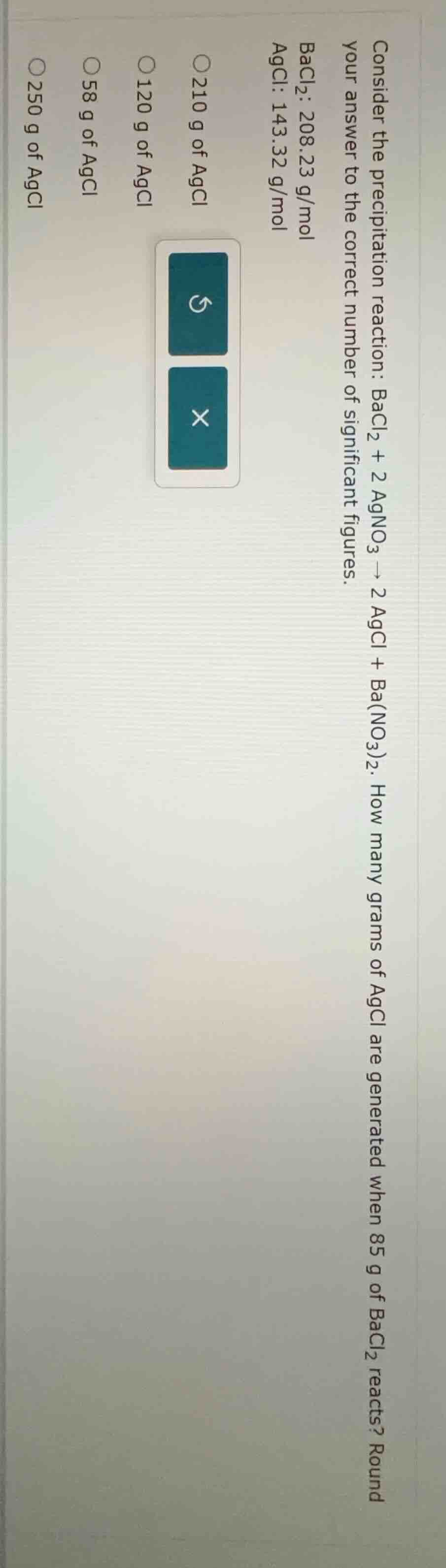
consider the precipitation reaction: bacl₂ + 2 agno₃ → 2 agcl + ba(no₃)₂. how many grams of agcl are generated when 85 g of bacl₂ reacts? round your answer to the correct number of significant figures. bacl₂: 208.23 g/mol agcl: 143.32 g/mol 210 g of agcl 120 g of agcl 58 g of agcl 250 g of agcl
Step1: Calculate moles of \( BaCl_2 \)
Moles of \( BaCl_2 = \frac{mass}{molar\ mass} = \frac{85\ g}{208.23\ g/mol} \approx 0.4082\ mol \)
Step2: Determine moles of \( AgCl \) from stoichiometry
From the reaction \( BaCl_2 + 2AgNO_3
ightarrow 2AgCl + Ba(NO_3)_2 \), 1 mol \( BaCl_2 \) produces 2 mol \( AgCl \). So moles of \( AgCl = 2 \times 0.4082\ mol = 0.8164\ mol \)
Step3: Calculate mass of \( AgCl \)
Mass of \( AgCl = moles \times molar\ mass = 0.8164\ mol \times 143.32\ g/mol \approx 117\ g \approx 120\ g \) (considering significant figures)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
120 g of AgCl (Option: 120 g of AgCl)