QUESTION IMAGE
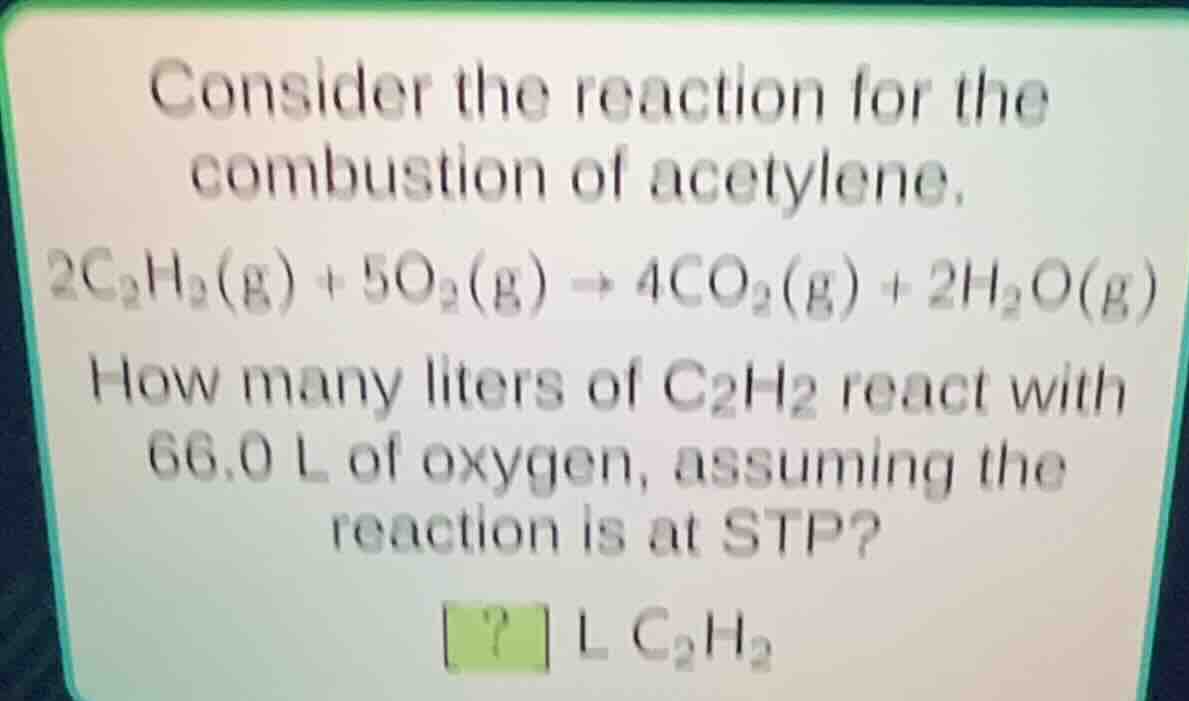
Question
consider the reaction for the combustion of acetylene.\\(2\ce{c_{2}h_{2}(g)} + 5\ce{o_{2}(g)} \
ightarrow 4\ce{co_{2}(g)} + 2\ce{h_{2}o(g)}\\)\
how many liters of \\(\ce{c_{2}h_{2}}\\) react with 66.0 l of oxygen, assuming the reaction is at stp?\
? l \\(\ce{c_{2}h_{2}}\\)
Step1: Relate gas volumes via mole ratio
At STP, gas volume ratio = mole ratio. From the reaction, $\frac{V_{\text{C}_2\text{H}_2}}{V_{\text{O}_2}} = \frac{2}{5}$
Step2: Solve for $V_{\text{C}_2\text{H}_2}$
Substitute $V_{\text{O}_2}=66.0\ \text{L}$ into the ratio.
$V_{\text{C}_2\text{H}_2} = 66.0\ \text{L} \times \frac{2}{5}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$26.4$ L $\text{C}_2\text{H}_2$