QUESTION IMAGE
Question
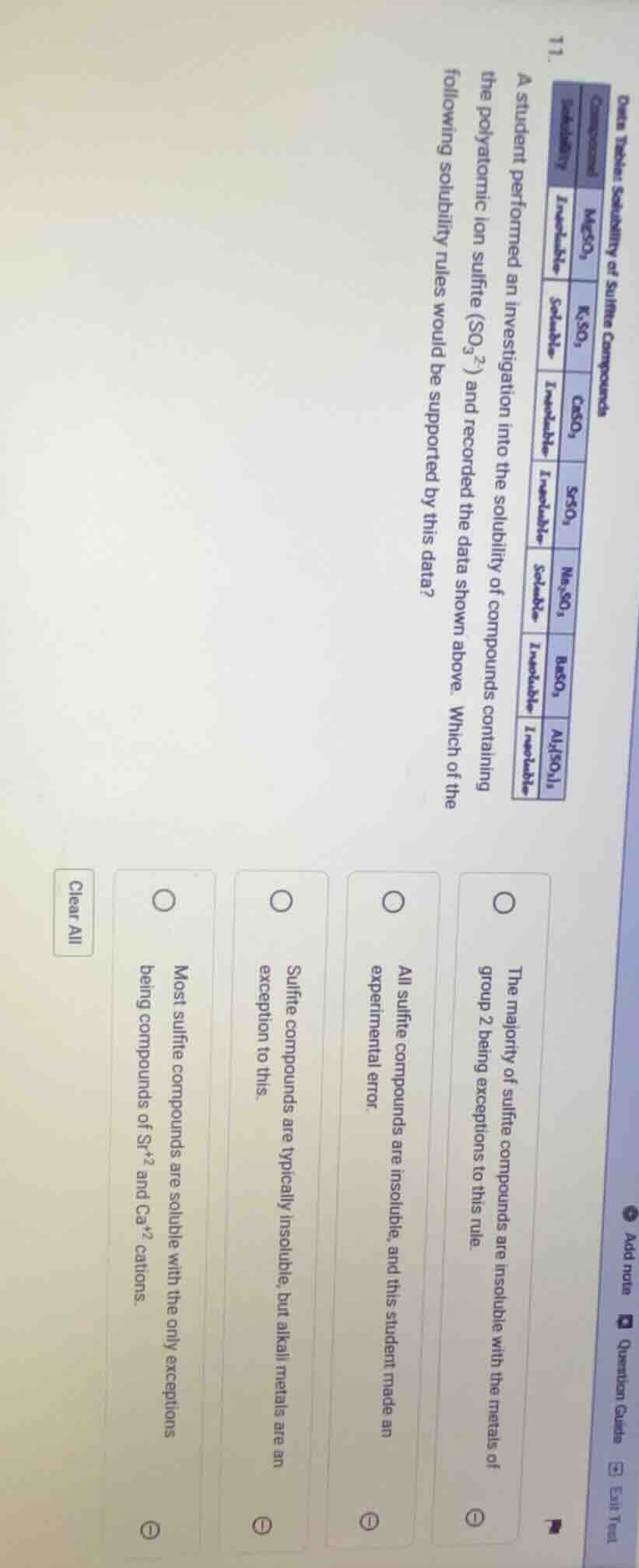
data table: solubility of sulfite compounds
11.
a student performed an investigation into the solubility of compounds containing the polyatomic ion sulfite (so₃²⁻) and recorded the data shown above. which of the following solubility rules would be supported by this data?
options:
the majority of sulfite compounds are insoluble with the metals of group 2 being exceptions to this rule.
all sulfite compounds are insoluble, and this student made an experimental error.
sulfite compounds are typically insoluble, but alkali metals are an exception to this.
most sulfite compounds are soluble with the only exceptions being compounds of sr²⁺ and ca²⁺ cations.
First, analyze the data: soluble sulfites are $K_2SO_3$, $Na_2SO_3$ (alkali metal cations), while insoluble ones are $MgSO_3$, $CaSO_3$, $SrSO_3$, $BaSO_3$, $Al_2(SO_3)_3$ (non-alkali metal cations). This matches the pattern that sulfites are mostly insoluble, with alkali metals as exceptions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Sulfite compounds are typically insoluble, but alkali metals are an exception to this.