QUESTION IMAGE
Question
date:
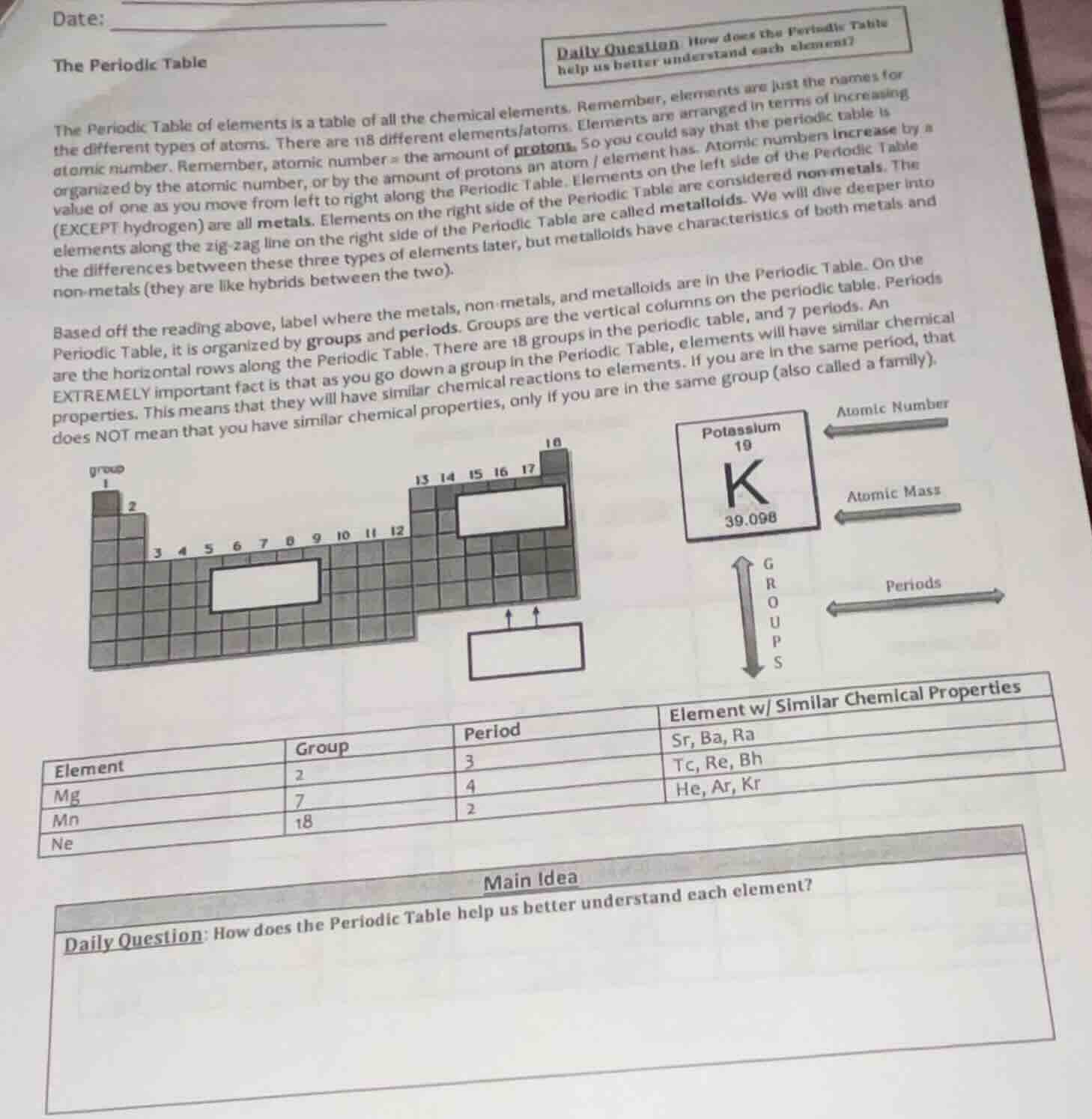
the periodic table
daily question: how does the periodic table
help us better understand each element?
the periodic table of elements is a table of all the chemical elements. remember, elements are just the names for
the different types of atoms. there are 118 different elements/atoms. elements are arranged in terms of increasing
atomic number. remember, atomic number = the amount of protons. so you could say that the periodic table is
organized by the atomic number, or by the amount of protons an atom / element has. atomic numbers increase by a
value of one as you move from left to right along the periodic table. elements on the left side of the periodic table
(except hydrogen) are all metals. elements on the right side of the periodic table are considered non - metals. the
elements along the zig - zag line on the right side of the periodic table are called metalloids. we will dive deeper into
the differences between these three types of elements later, but metalloids have characteristics of both metals and
non - metals (they are like hybrids between the two).
based off the reading above, label where the metals, non - metals, and metalloids are in the periodic table. on the
periodic table, it is organized by groups and periods. groups are the vertical columns on the periodic table. periods
are the horizontal rows along the periodic table. there are 18 groups in the periodic table, and 7 periods. an
extremely important fact is that as you go down a group in the periodic table, elements will have similar chemical
properties. this means that they will have similar chemical reactions to elements. if you are in the same period, that
does not mean that you have similar chemical properties, only if you are in the same group (also called a family).
| element | group | period | element w/ similar chemical properties |
|---|---|---|---|
| mn | 7 | 4 | tc, re, bh |
| ne | 18 | 2 | he, ar, kr |
main idea
daily question: how does the periodic table help us better understand each element?
To answer the daily question "How does the Periodic Table help us better understand each element?", we analyze the information from the reading:
- Organization by Atomic Number: The Periodic Table arranges elements by increasing atomic number (number of protons), so we can quickly identify an element’s atomic structure.
- Classification of Elements: It groups elements into metals (left, except H), non - metals (right), and metalloids (zig - zag line), helping us understand their general physical/chemical properties.
- Group and Period Trends: Elements in the same group (vertical column) have similar chemical properties (e.g., Mg in Group 2 has properties like Sr, Ba, Ra). Periods (horizontal rows) show trends in atomic properties as atomic number increases.
- Predicting Properties: By an element’s position (group/period), we can predict its chemical reactions (e.g., Group 18 noble gases like Ne are unreactive, similar to He, Ar, Kr).
The Periodic Table helps us understand elements by organizing them by atomic number, classifying them (metals, non - metals, metalloids), showing group - based chemical property similarities, and allowing property prediction from position.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The Periodic Table helps us better understand each element by:
- Organizing elements by atomic number (proton count) to show atomic structure.
- Classifying elements as metals (left, except H), non - metals (right), or metalloids (zig - zag) to indicate general properties.
- Grouping elements (vertical columns) with similar chemical properties (e.g., same - group elements react similarly).
- Using periods (horizontal rows) to show trends in properties as atomic number increases, and enabling property prediction from an element’s position.