QUESTION IMAGE
Question
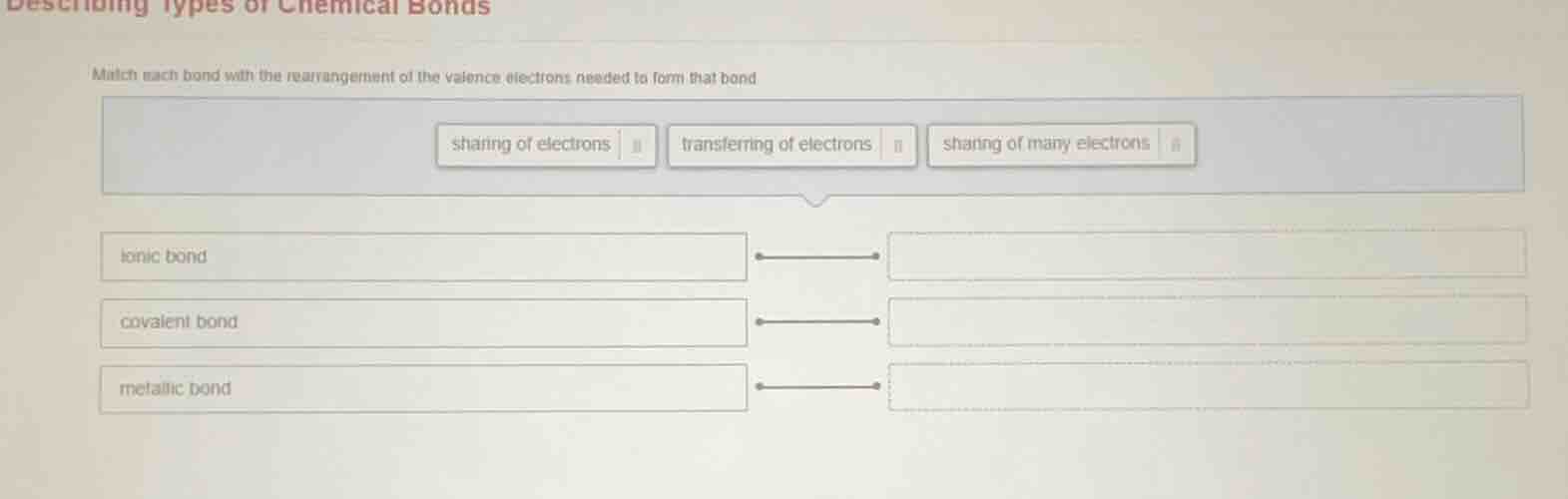
describing types of chemical bonds
match each bond with the rearrangement of the valence electrons needed to form that bond
sharing of electrons
transferring of electrons
sharing of many electrons
ionic bond
covalent bond
metallic bond
Brief Explanations
- Ionic bonds form when electrons are transferred between atoms, creating charged ions that attract each other.
- Covalent bonds form when atoms share valence electrons to achieve a stable electron configuration.
- Metallic bonds involve a "sea" of delocalized electrons shared among many metal atoms, which is described as sharing of many electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Ionic bond: transferring of electrons
- Covalent bond: sharing of electrons
- Metallic bond: sharing of many electrons