QUESTION IMAGE
Question
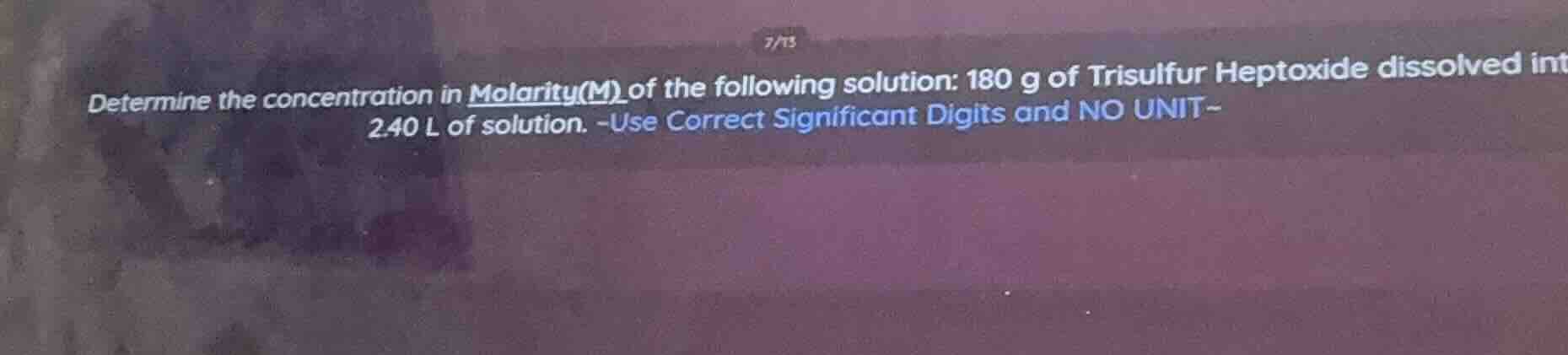
determine the concentration in molarity(m) of the following solution: 180 g of trisulfur heptoxide dissolved int 2.40 l of solution. -use correct significant digits and no unit-
Step1: Find molar mass of $S_3O_7$
Molar mass of S = 32.07 g/mol, O = 16.00 g/mol
$$\text{Molar mass} = (3 \times 32.07) + (7 \times 16.00) = 96.21 + 112.00 = 208.21\ \text{g/mol}$$
Step2: Calculate moles of solute
$$\text{Moles} = \frac{\text{Mass}}{\text{Molar mass}} = \frac{180}{208.21} \approx 0.8645\ \text{mol}$$
Step3: Compute molarity
$$\text{Molarity} = \frac{\text{Moles of solute}}{\text{Volume of solution (L)}} = \frac{0.8645}{2.40} \approx 0.360$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.360