QUESTION IMAGE
Question
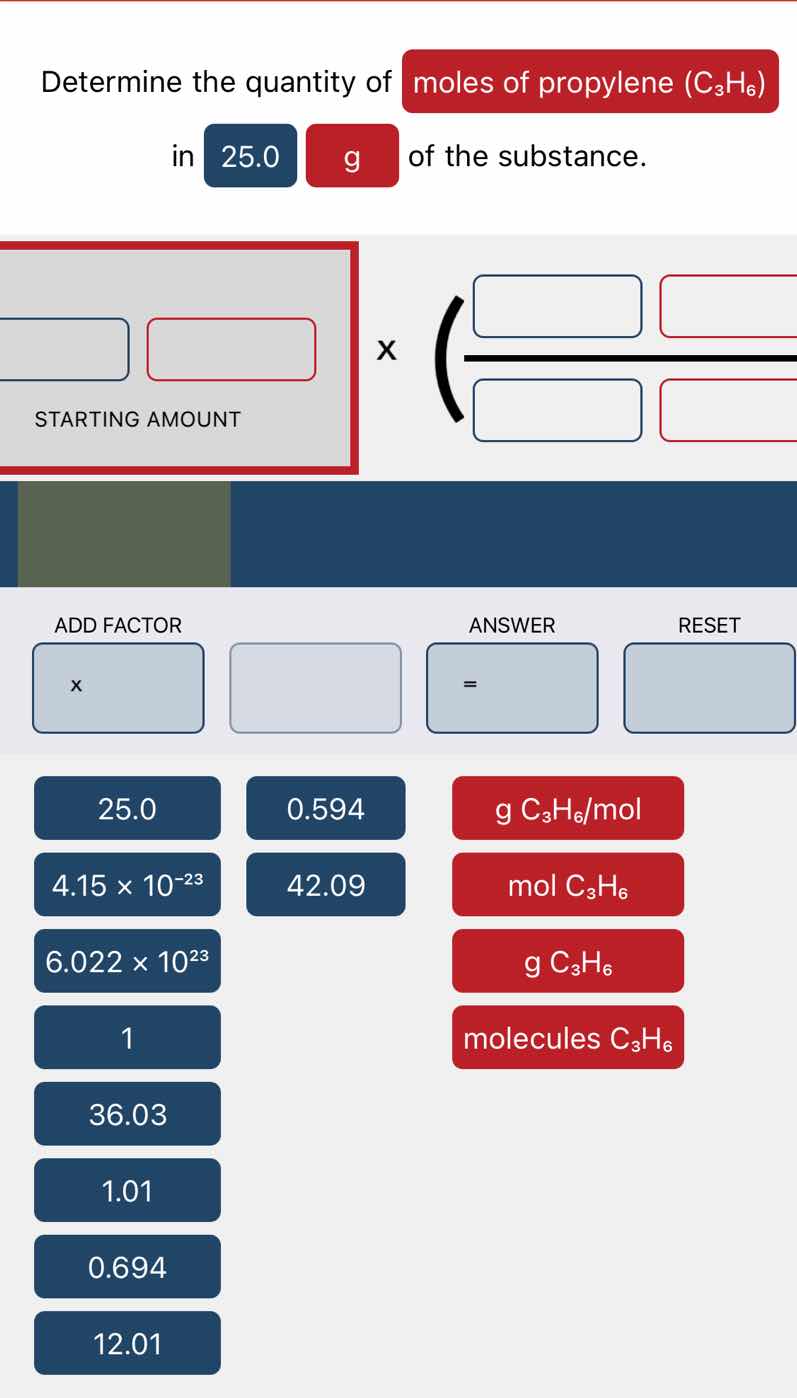
determine the quantity of moles of propylene (c₃h₆) in 25.0 g of the substance. starting amount x ( / ) add factor answer reset x = 25.0 0.594 g c₃h₆/mol 4.15 × 10⁻²³ 42.09 mol c₃h₆ 6.022 × 10²³ g c₃h₆ 1 molecules c₃h₆ 36.03 1.01 0.694 12.01
Step1: Identify starting amount
Starting amount: $25.0\ \text{g}\ \text{C}_3\text{H}_6$
Step2: Calculate molar mass of $\text{C}_3\text{H}_6$
Molar mass = $(3 \times 12.01) + (6 \times 1.01) = 42.09\ \text{g}\ \text{C}_3\text{H}_6/\text{mol}$
Step3: Convert mass to moles
Use conversion factor $\frac{1\ \text{mol}\ \text{C}_3\text{H}_6}{42.09\ \text{g}\ \text{C}_3\text{H}_6}$
Expression: $25.0\ \text{g}\ \text{C}_3\text{H}_6 \times \frac{1\ \text{mol}\ \text{C}_3\text{H}_6}{42.09\ \text{g}\ \text{C}_3\text{H}_6}$
Step4: Compute final value
Calculate: $\frac{25.0}{42.09} = 0.594$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$0.594\ \text{mol}\ \text{C}_3\text{H}_6$
(Note: Filling the interactive template:
Starting amount: $25.0$, unit: $\text{g}\ \text{C}_3\text{H}_6$
Conversion factor numerator: $1$, unit: $\text{mol}\ \text{C}_3\text{H}_6$
Conversion factor denominator: $42.09$, unit: $\text{g}\ \text{C}_3\text{H}_6$)