QUESTION IMAGE
Question
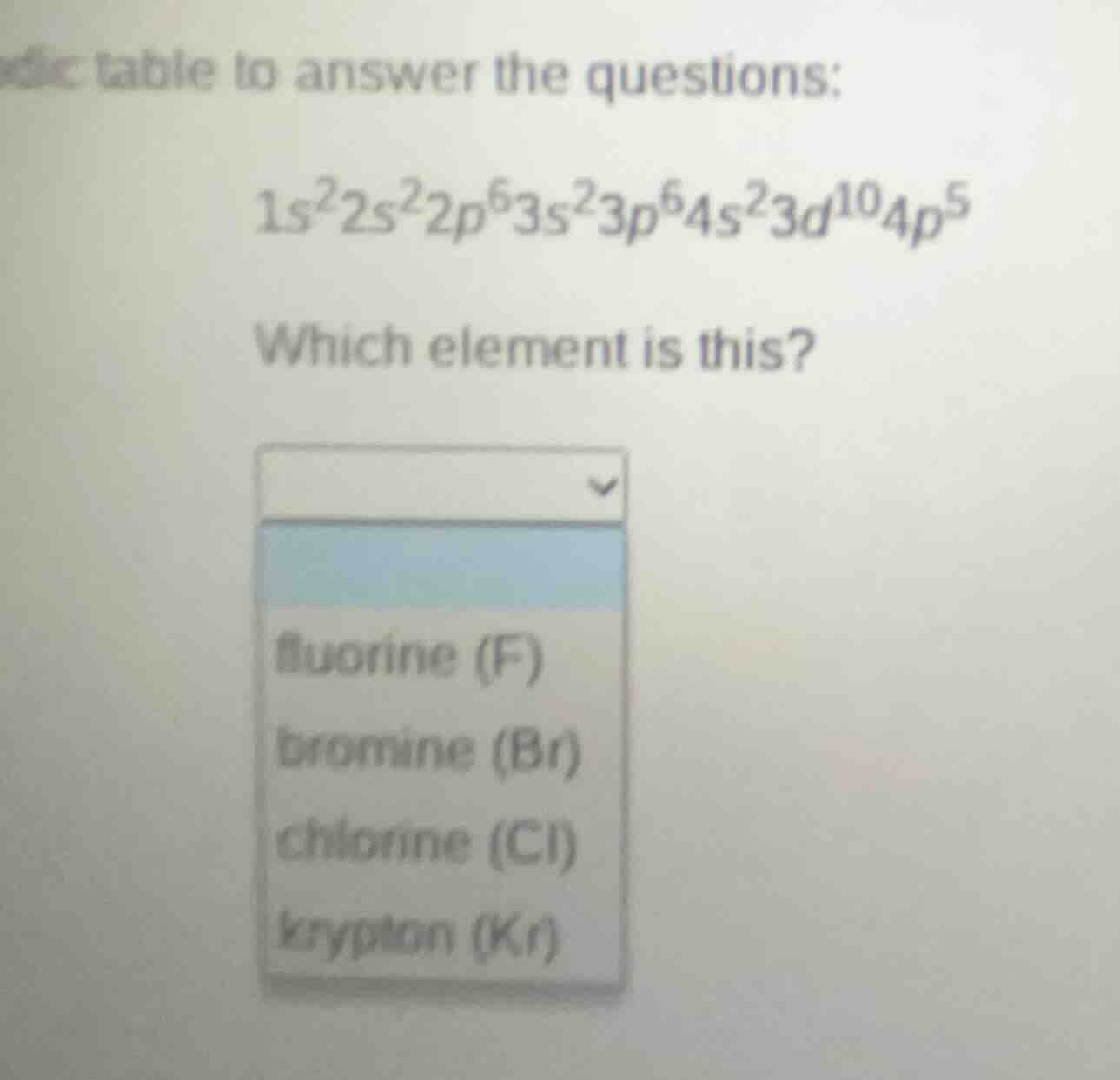
dic table to answer the questions:
$1s^22s^22p^63s^23p^64s^23d^{10}4p^5$
which element is this?
fluorine (f)
bromine (br)
chlorine (cl)
krypton (kr)
Step1: Calculate total electrons
Sum the exponents in the electron configuration: \(2 + 2 + 6 + 2 + 6 + 2 + 10 + 5 = 35\).
Step2: Identify element by atomic number
The atomic number equals the number of electrons (for a neutral atom). Bromine (Br) has an atomic number of 35. Fluorine (F) has atomic number 9, chlorine (Cl) has 17, and krypton (Kr) has 36.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. bromine (Br)