QUESTION IMAGE
Question
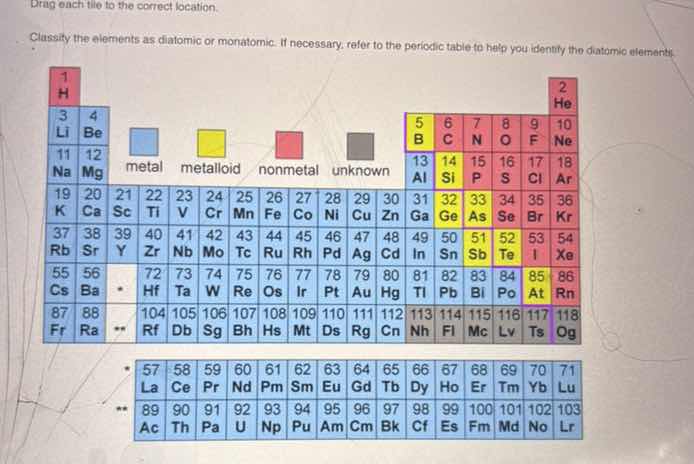
drag each tile to the correct location.
classify the elements as diatomic or monatomic. if necessary, refer to the periodic table to help you identify the diatomic elements.
To solve this, we first recall the definitions:
- Monatomic elements: Exist as single atoms (most elements, like metals, noble gases, some nonmetals).
- Diatomic elements: Exist as molecules with 2 atoms (e.g., \( \text{H}_2, \text{N}_2, \text{O}_2, \text{F}_2, \text{Cl}_2, \text{Br}_2, \text{I}_2 \); also \( \text{C} \) (diamond/graphite) is monatomic, but diatomic \( \text{C}_2 \) is rare).
Step 1: Identify Diatomic Elements
From the periodic table, the diatomic elements (common) are:
- Hydrogen (H): Diatomic (\( \text{H}_2 \))
- Nitrogen (N): Diatomic (\( \text{N}_2 \))
- Oxygen (O): Diatomic (\( \text{O}_2 \))
- Fluorine (F): Diatomic (\( \text{F}_2 \))
- Chlorine (Cl): Diatomic (\( \text{Cl}_2 \))
- Bromine (Br): Diatomic (\( \text{Br}_2 \))
- Iodine (I): Diatomic (\( \text{I}_2 \))
Step 2: Identify Monatomic Elements
Most other elements are monatomic (exist as single atoms):
- Metals: Li, Be, Na, Mg, K, Ca, etc. (all elements in metal/metalloid/nonmetal/unknown blocks except diatomic ones).
- Noble Gases: He, Ne, Ar, Kr, Xe, Og (monatomic, as \( \text{He}, \text{Ne} \), etc.).
- Nonmetals/Metalloids: B, C, Si, P, S, As, Se, Sb, Te, At, etc. (except diatomic nonmetals like H, N, O, F, Cl, Br, I).
Classification:
- Diatomic: H, N, O, F, Cl, Br, I
- Monatomic: All others (e.g., He, Li, Be, B, C, Si, P, S, Ar, K, Ca, etc.)
For example, if tiles include:
- H, N, O, F, Cl, Br, I → Diatomic
- He, Li, Be, B, C, Si, P, S, Ar, K, Ca → Monatomic
(Note: If specific tiles are provided, drag diatomic elements to "diatomic" and monatomic to "monatomic".)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we first recall the definitions:
- Monatomic elements: Exist as single atoms (most elements, like metals, noble gases, some nonmetals).
- Diatomic elements: Exist as molecules with 2 atoms (e.g., \( \text{H}_2, \text{N}_2, \text{O}_2, \text{F}_2, \text{Cl}_2, \text{Br}_2, \text{I}_2 \); also \( \text{C} \) (diamond/graphite) is monatomic, but diatomic \( \text{C}_2 \) is rare).
Step 1: Identify Diatomic Elements
From the periodic table, the diatomic elements (common) are:
- Hydrogen (H): Diatomic (\( \text{H}_2 \))
- Nitrogen (N): Diatomic (\( \text{N}_2 \))
- Oxygen (O): Diatomic (\( \text{O}_2 \))
- Fluorine (F): Diatomic (\( \text{F}_2 \))
- Chlorine (Cl): Diatomic (\( \text{Cl}_2 \))
- Bromine (Br): Diatomic (\( \text{Br}_2 \))
- Iodine (I): Diatomic (\( \text{I}_2 \))
Step 2: Identify Monatomic Elements
Most other elements are monatomic (exist as single atoms):
- Metals: Li, Be, Na, Mg, K, Ca, etc. (all elements in metal/metalloid/nonmetal/unknown blocks except diatomic ones).
- Noble Gases: He, Ne, Ar, Kr, Xe, Og (monatomic, as \( \text{He}, \text{Ne} \), etc.).
- Nonmetals/Metalloids: B, C, Si, P, S, As, Se, Sb, Te, At, etc. (except diatomic nonmetals like H, N, O, F, Cl, Br, I).
Classification:
- Diatomic: H, N, O, F, Cl, Br, I
- Monatomic: All others (e.g., He, Li, Be, B, C, Si, P, S, Ar, K, Ca, etc.)
For example, if tiles include:
- H, N, O, F, Cl, Br, I → Diatomic
- He, Li, Be, B, C, Si, P, S, Ar, K, Ca → Monatomic
(Note: If specific tiles are provided, drag diatomic elements to "diatomic" and monatomic to "monatomic".)