QUESTION IMAGE
Question
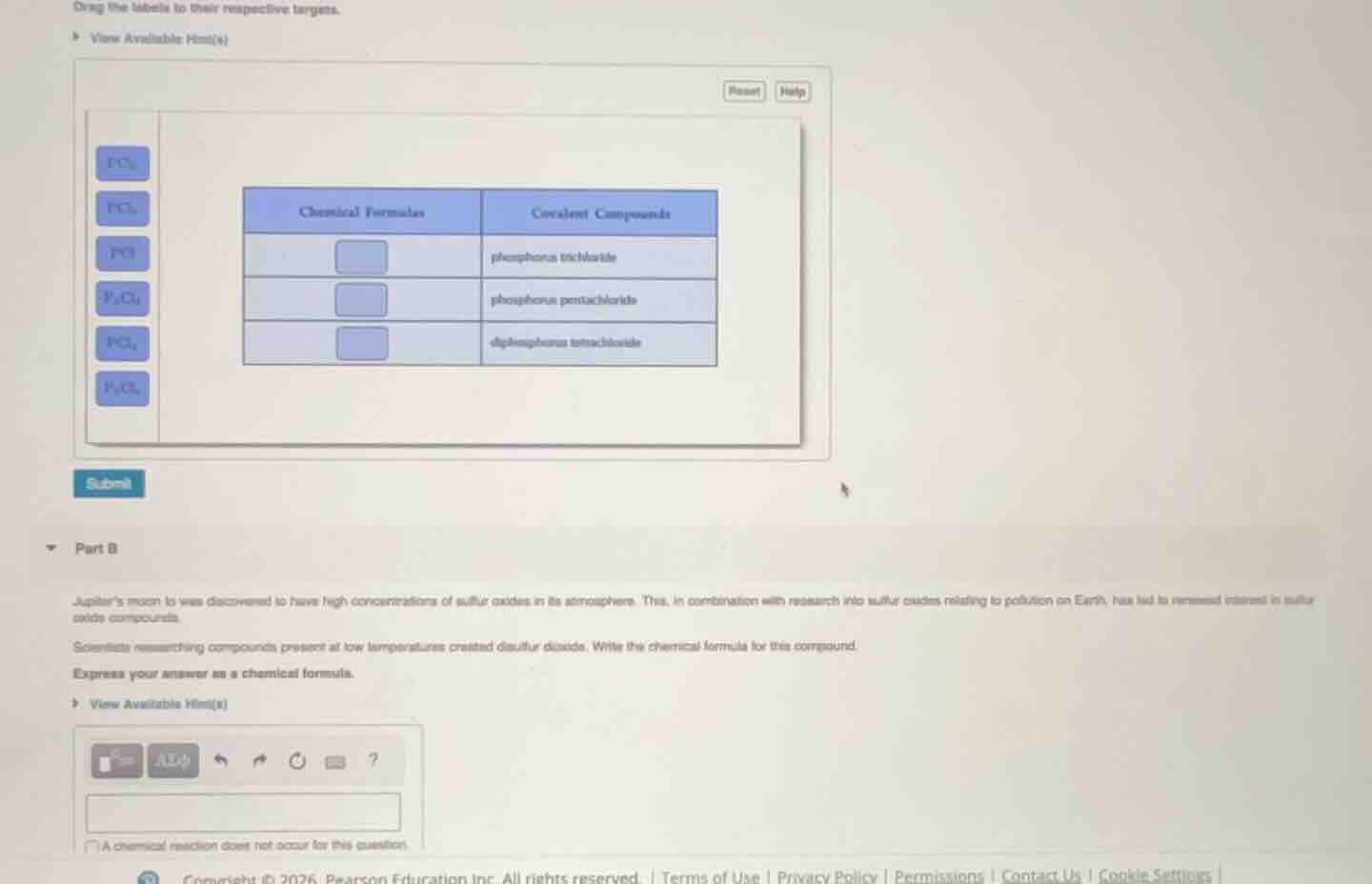
drag the labels to their respective targets.
view available hint(s)
reset help
chemical formulas covalent compounds
phosphorus trichloride
phosphorus pentachloride
diphosphorus tetrachloride
submit
part b
jupiter’s moon io was discovered to have high concentrations of sulfur oxides in its atmosphere. this, in combination with research into sulfur oxides relating to pollution on earth, has led to renewed interest in sulfur oxide compounds.
scientists researching compounds present at low temperatures created disulfur dioxide. write the chemical formula for this compound.
express your answer as a chemical formula.
view available hint(s)
a chemical reaction does not occur for this question.
copyright © 2026 pearson education inc. all rights reserved. | terms of use | privacy policy | permissions | contact us | cookie settings
Part A (Matching Chemical Formulas to Covalent Compounds)
Step 1: Analyze Phosphorus Trichloride
Phosphorus trichloride has one phosphorus (P) and three chlorine (Cl) atoms. So its formula is $PCl_3$.
Step 2: Analyze Phosphorus Pentachloride
Phosphorus pentachloride has one phosphorus (P) and five chlorine (Cl) atoms. So its formula is $PCl_5$.
Step 3: Analyze Diphosphorus Tetrachloride
Diphosphorus tetrachloride has two phosphorus (P) and four chlorine (Cl) atoms. So its formula is $P_2Cl_4$.
Part B (Chemical Formula for Disulfur Dioxide)
Step 1: Identify Atoms and Prefixes
"Disulfur" means 2 sulfur (S) atoms, and "dioxide" means 2 oxygen (O) atoms.
Step 2: Write the Formula
Combine the atoms with their respective counts. So the formula is $S_2O_2$.
Part A Answers:
- Phosphorus trichloride: $PCl_3$
- Phosphorus pentachloride: $PCl_5$
- Diphosphorus tetrachloride: $P_2Cl_4$
Part B Answer:
$S_2O_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Step 1: Identify Atoms and Prefixes
"Disulfur" means 2 sulfur (S) atoms, and "dioxide" means 2 oxygen (O) atoms.
Step 2: Write the Formula
Combine the atoms with their respective counts. So the formula is $S_2O_2$.
Part A Answers:
- Phosphorus trichloride: $PCl_3$
- Phosphorus pentachloride: $PCl_5$
- Diphosphorus tetrachloride: $P_2Cl_4$
Part B Answer:
$S_2O_2$