QUESTION IMAGE
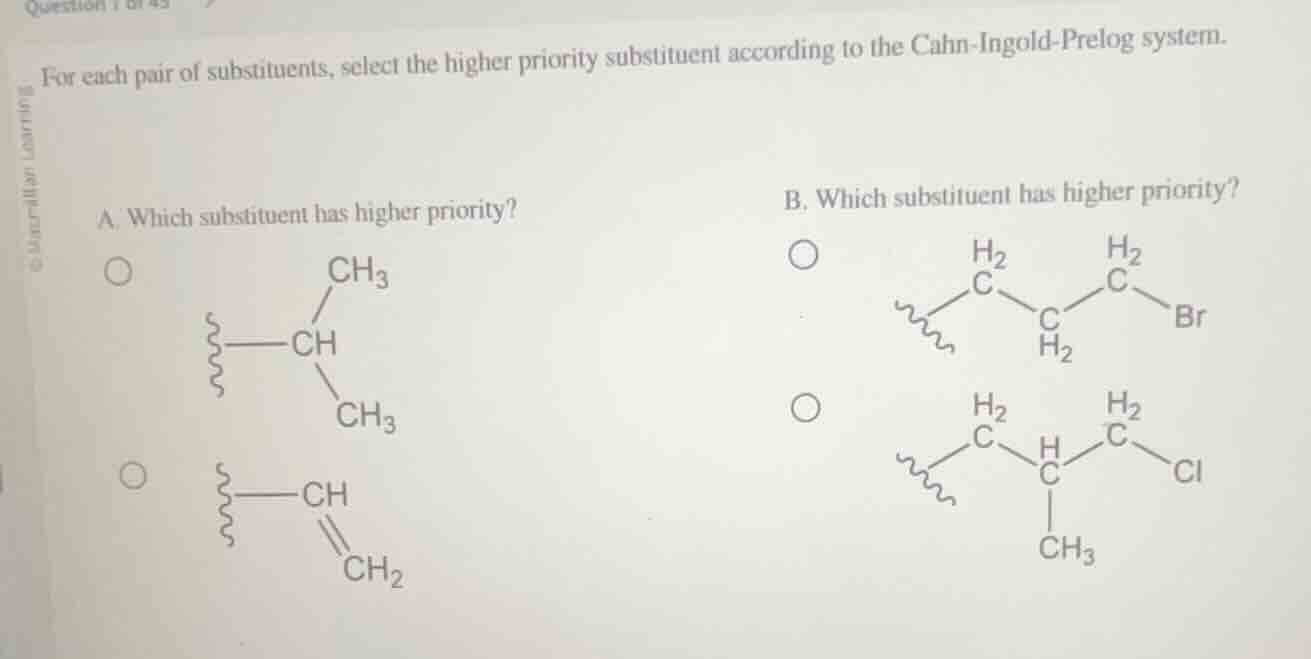
Question
for each pair of substituents, select the higher priority substituent according to the cahn-ingold-prelog system.
a. which substituent has higher priority?
$ce{-ch(ch3)2}$
$ce{-ch=ch2}$
b. which substituent has higher priority?
$ce{-ch2ch2ch2br}$
$ce{-ch2ch(ch3)ch2cl}$
For part A: Using Cahn-Ingold-Prelog rules, first compare directly attached atoms (both C here). Then look at the next bonded atoms: the isopropyl group ($\text{-CH(CH}_3\text{)}_2$) has C bonded to two C and one H, while the vinyl group ($\text{-CH=CH}_2$) has C bonded to two C (via double bond, counted as two single bonds) and one H. The double bond means the vinyl group's C has higher effective substitution priority.
For part B: Compare the first point of difference. The top substituent has a terminal Br, the bottom has a terminal Cl, but the first branching point on the bottom is a C bonded to a methyl. At the second carbon from the attachment, the top has a CH₂, the bottom has a C bonded to CH₃ and CH₂CH₂Cl. But Br has a higher atomic number than Cl, and we compare the highest atomic number atoms at the first point of difference. Wait, no: for the bottom substituent, the second carbon (attached to the first CH₂) is bonded to CH₃, CH₂, and CH₂CH₂Cl. For the top, the second carbon is CH₂ bonded to CH₂Br. The first point where atomic numbers differ: the bottom's second carbon has a C (from CH₃) vs the top's second carbon has H? No, wait, the attachment point is the first CH₂ for both. First attached atom is C for both. Next, top's C is bonded to H, H, C (CH₂CH₂Br); bottom's C is bonded to H, H, C (CH(CH₃)CH₂Cl). Now look at the next level: top's next C is bonded to H, H, Br; bottom's next C is bonded to CH₃, H, CH₂Cl. Br has atomic number 35, which is higher than Cl (17), so the top substituent has higher priority.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $\boldsymbol{\ce{-CH=CH2}}$ (the vinyl substituent)
B. $\boldsymbol{\ce{-CH2CH2CH2Br}}$ (the terminal bromine-containing substituent)