QUESTION IMAGE
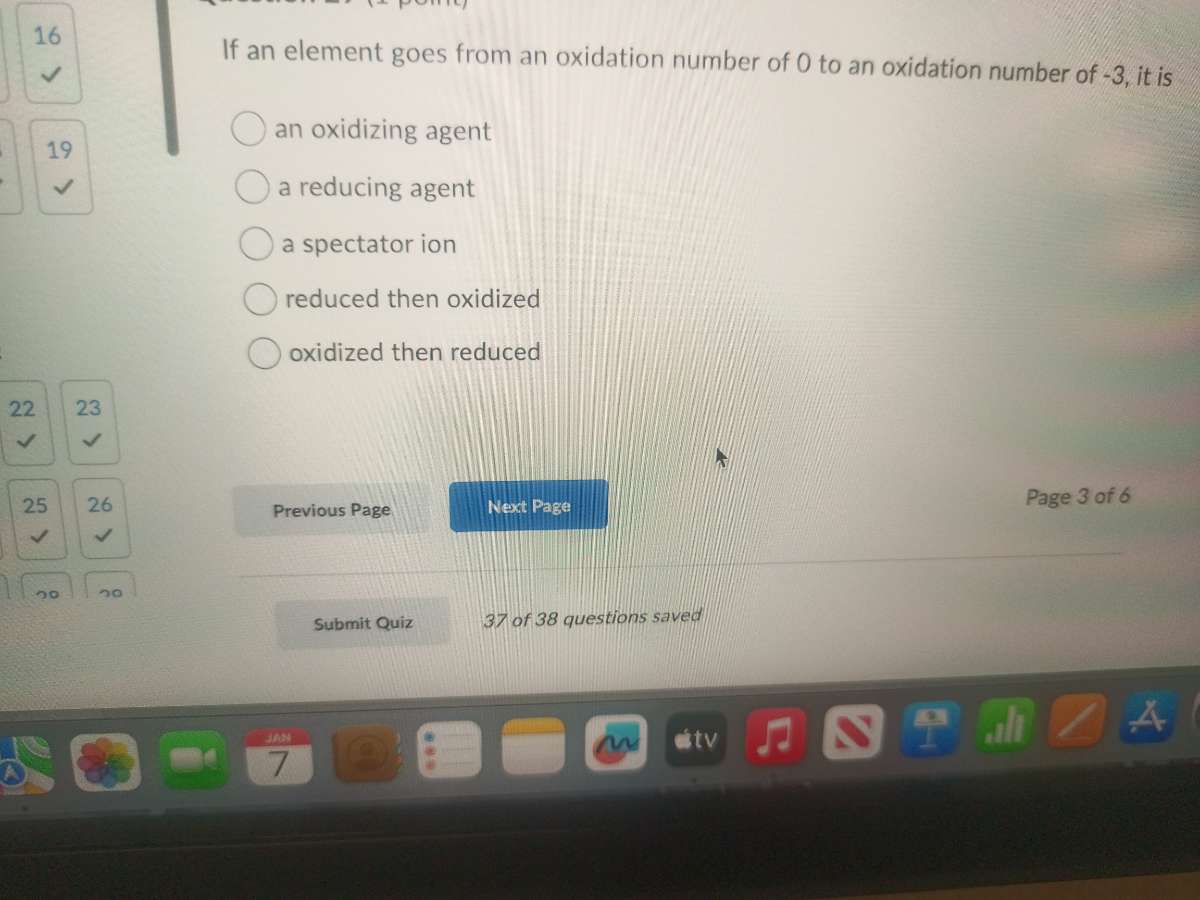
Question
if an element goes from an oxidation number of 0 to an oxidation number of -3, it is
an oxidizing agent
a reducing agent
a spectator ion
reduced then oxidized
oxidized then reduced
To determine the role of the element, we analyze the change in oxidation number. An oxidation number decrease (from 0 to -3) means the element gains electrons, which is reduction. An oxidizing agent is reduced (since it causes another substance to be oxidized by accepting electrons). A reducing agent is oxidized (loses electrons). A spectator ion doesn't change oxidation state. The element's oxidation number only decreases, so it's reduced, acting as an oxidizing agent.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. an oxidizing agent