QUESTION IMAGE
Question
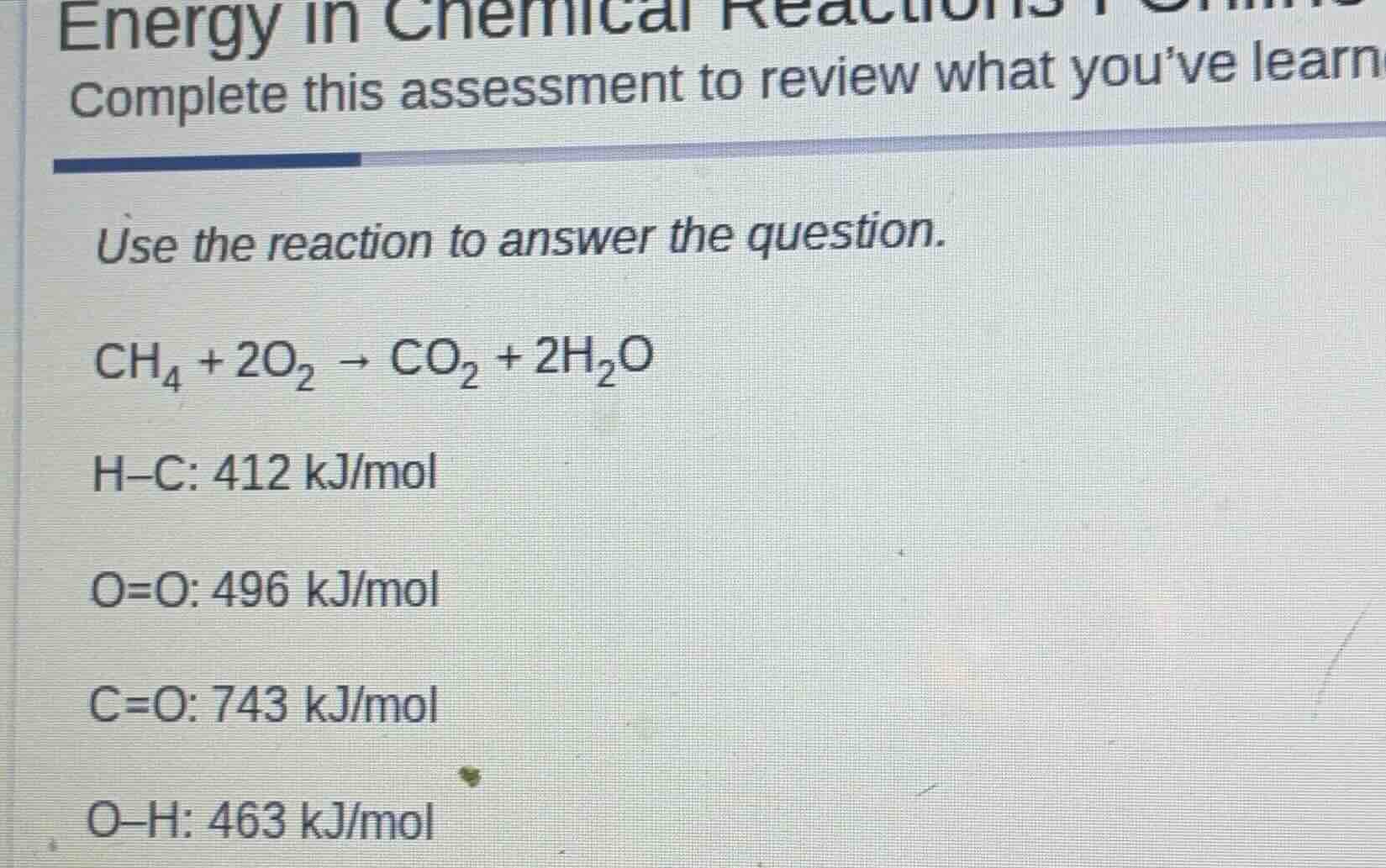
energy in chemical reactions complete this assessment to review what you’ve learn use the reaction to answer the question. \\(\ce{ch_{4} + 2o_{2} \
ightarrow co_{2} + 2h_{2}o}\\) \\(\ce{h-c}\\): 412 kj/mol \\(\ce{o=o}\\): 496 kj/mol \\(\ce{c=o}\\): 743 kj/mol \\(\ce{o-h}\\): 463 kj/mol
Assuming the question is to calculate the enthalpy change ($\Delta H$) of the reaction using bond energies (since it's a common problem with this setup), here's the solution:
Step1: Calculate energy required to break bonds (reactants)
- In $\ce{CH4}$, there are 4 $\ce{H-C}$ bonds. Energy for breaking 4 $\ce{H-C}$: $4\times412\ \text{kJ/mol} = 1648\ \text{kJ/mol}$
- In $2\ce{O2}$, there are 2 $\ce{O=O}$ bonds. Energy for breaking 2 $\ce{O=O}$: $2\times496\ \text{kJ/mol} = 992\ \text{kJ/mol}$
- Total energy to break bonds: $1648 + 992 = 2640\ \text{kJ/mol}$
Step2: Calculate energy released when forming bonds (products)
- In $\ce{CO2}$, there are 2 $\ce{C=O}$ bonds. Energy for forming 2 $\ce{C=O}$: $2\times743\ \text{kJ/mol} = 1486\ \text{kJ/mol}$
- In $2\ce{H2O}$, each $\ce{H2O}$ has 2 $\ce{O-H}$ bonds, so 4 $\ce{O-H}$ bonds total. Energy for forming 4 $\ce{O-H}$: $4\times463\ \text{kJ/mol} = 1852\ \text{kJ/mol}$
- Total energy released: $1486 + 1852 = 3338\ \text{kJ/mol}$
Step3: Calculate $\Delta H$ (enthalpy change)
$\Delta H = \text{Energy to break bonds} - \text{Energy released to form bonds}$
$\Delta H = 2640 - 3338 = -698\ \text{kJ/mol}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The enthalpy change of the reaction is $\boldsymbol{-698\ \text{kJ/mol}}$ (indicating an exothermic reaction).