QUESTION IMAGE
Question
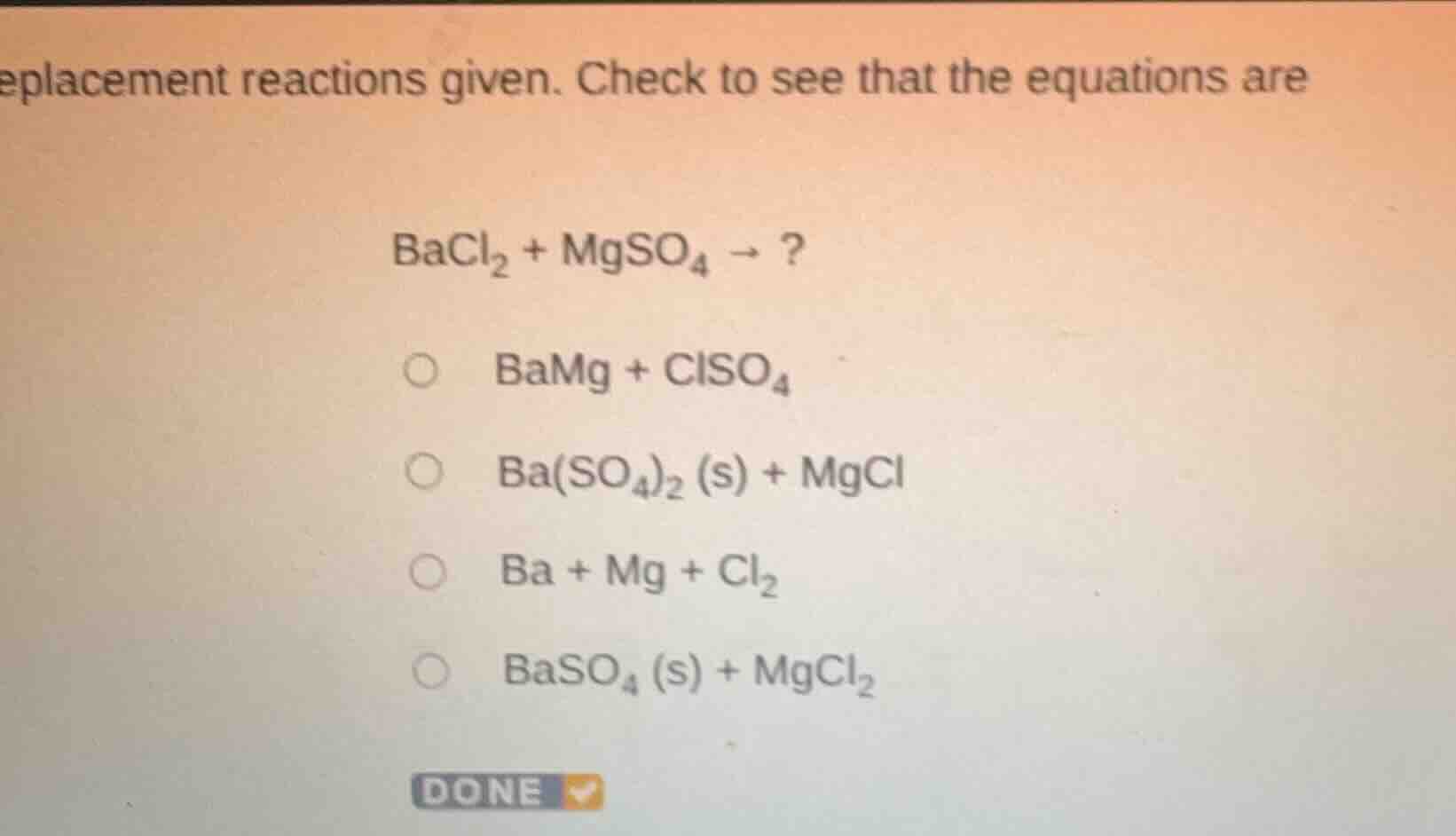
eplacement reactions given. check to see that the equations are bacl₂ + mgso₄ →? ○ bamg + clso₄ ○ ba(so₄)₂ (s) + mgcl ○ ba + mg + cl₂ ○ baso₄ (s) + mgcl₂
Step1: Identify reaction type
This is a double - replacement reaction. In a double - replacement reaction, the cations and anions of the two reactants switch places. The general form is \(AB + CD
ightarrow AD+CB\). For the reaction \(BaCl_{2}+MgSO_{4}\), the cation of \(BaCl_{2}\) is \(Ba^{2 + }\), anion is \(Cl^{-}\); the cation of \(MgSO_{4}\) is \(Mg^{2+}\), anion is \(SO_{4}^{2 - }\).
Step2: Determine products
When we switch the cations and anions, the new compounds formed should be \(BaSO_{4}\) (since \(Ba^{2+}\) combines with \(SO_{4}^{2 - }\)) and \(MgCl_{2}\) (since \(Mg^{2+}\) combines with \(Cl^{-}\)). Barium sulfate (\(BaSO_{4}\)) is insoluble in water, so it will be a solid (\(s\)). Let's check the other options:
- Option 1: \(BaMg + ClSO_{4}\) is not a valid chemical formula as the combination of elements is incorrect.
- Option 2: The formula for barium sulfate should be \(BaSO_{4}\) (not \(Ba(SO_{4})_{2}\)) and the formula for magnesium chloride should be \(MgCl_{2}\) (not \(MgCl\)).
- Option 3: This is not a double - replacement reaction product, it looks like a decomposition or incorrect combination.
- Option 4: \(BaSO_{4}(s)+MgCl_{2}\) follows the double - replacement reaction rule, with \(Ba^{2+}\) combining with \(SO_{4}^{2 - }\) to form insoluble \(BaSO_{4}\) and \(Mg^{2+}\) combining with \(Cl^{-}\) to form \(MgCl_{2}\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(BaSO_{4}(s)+MgCl_{2}\) (the last option: \(BaSO_{4}(s)+MgCl_{2}\))