QUESTION IMAGE
Question
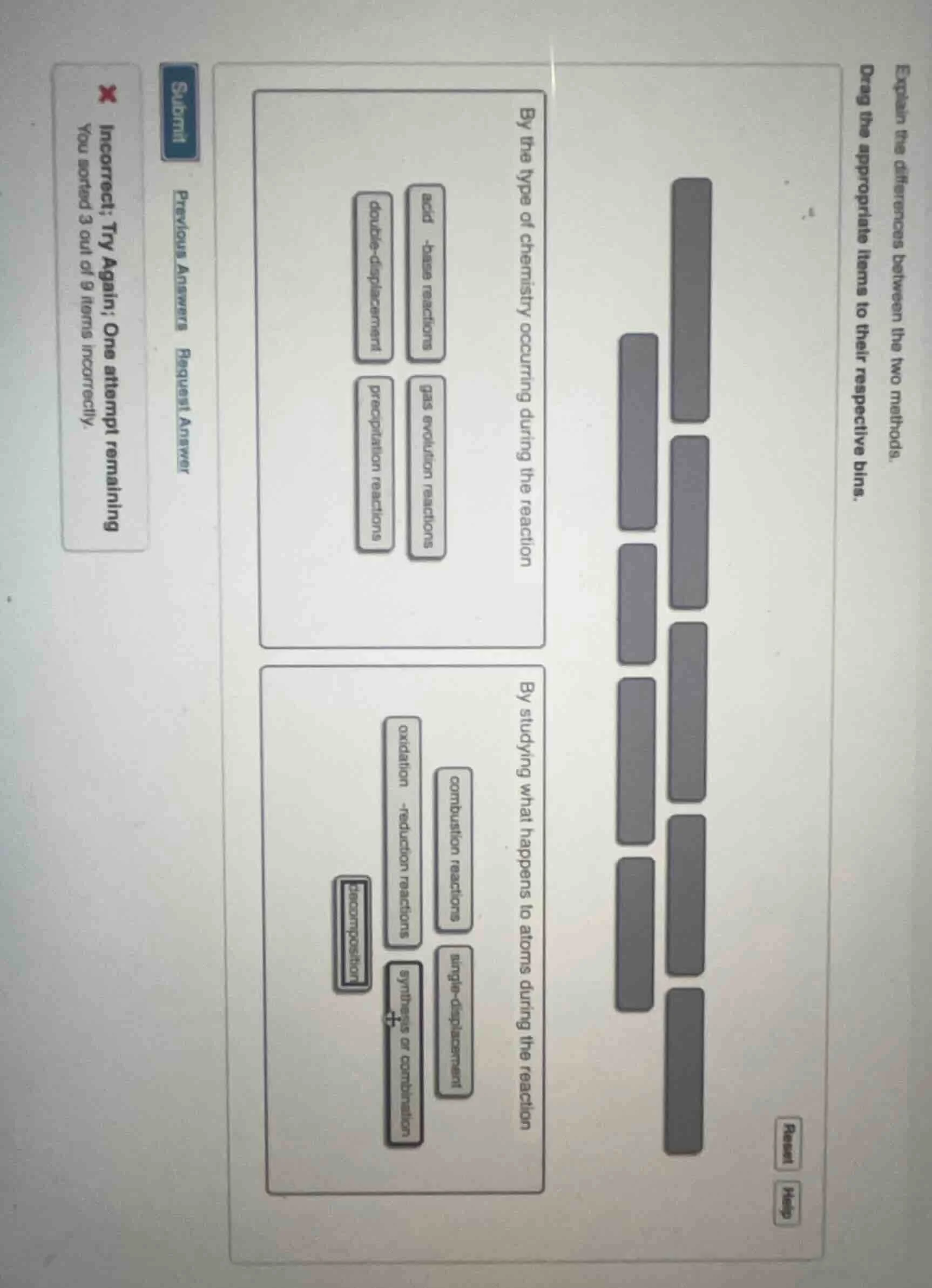
explain the differences between the two methods. drag the appropriate items to their respective bins.
by the type of chemistry occurring during the reaction
acid - base reactions
gas evolution reactions
double - displacement
precipitation reactions
by studying what happens to atoms during the reaction
combustion reactions
oxidation - reduction reactions
single - displacement
decomposition
synthesis or combination
incorrect; try again; one attempt remaining
you sorted 3 out of 9 items incorrectly.
submit
previous answers
request answer
reset
help
To solve this, we analyze the two methods of classifying chemical reactions:
- "By the type of chemistry occurring during the reaction" (focuses on observable changes or reaction types based on products/processes):
- Acid - base reactions (involve proton transfer, observable as neutralization, etc.).
- Double - displacement reactions (ions exchange partners, e.g., \(AB + CD
ightarrow AD + CB\)).
- Precipitation reactions (form a solid precipitate, observable).
- Gas evolution reactions (produce a gas, observable).
- "By studying what happens to atoms during the reaction" (focuses on electron/atom transfer, oxidation states, or fundamental reaction types):
- Oxidation - reduction reactions (involve electron transfer, change in oxidation states).
- Combustion reactions (a type of redox reaction, fuel + \(O_2\) → \(CO_2 + H_2O\) usually).
- Single - displacement reactions (one element displaces another, involves redox, e.g., \(A + BC
ightarrow AC + B\)).
- Decomposition (a compound breaks down, can be redox or not, e.g., \(AB
ightarrow A + B\)).
- Synthesis or combination (elements/compounds combine, e.g., \(A + B
ightarrow AB\), can be redox).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Bin 1: "By the type of chemistry occurring during the reaction"
- acid - base reactions
- double - displacement
- precipitation reactions
- gas evolution reactions
Bin 2: "By studying what happens to atoms during the reaction"
- oxidation - reduction reactions
- combustion reactions
- single - displacement
- decomposition
- synthesis or combination