QUESTION IMAGE
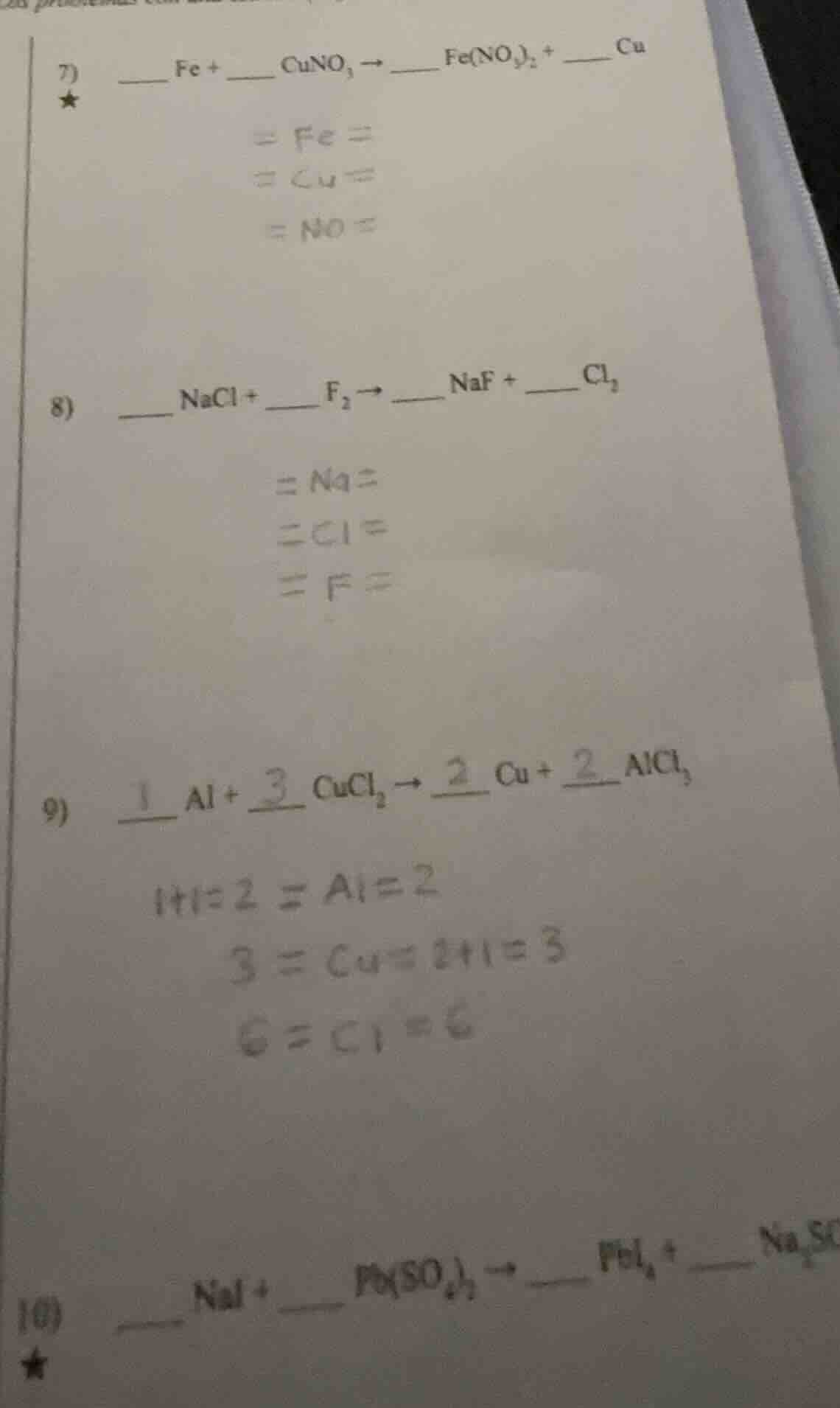
Question
- _ fe + _ cuno₃ → _ fe(no₃)₂ + _ cu
= fe =
= cu =
= no =
- _ nacl + _ f₂ → _ naf + _ cl₂
= na =
= cl =
= f =
- _ al + _ cucl₂ → _ cu + _ alcl₃
1+1=2 = al = 2
3 = cu = 2+1=3
6 = cl = 6
- _ nai + _ pb(so₄) → _ pbi₄ + _ na₂so
Problem 7: Balancing \( \boldsymbol{\text{Fe} + \text{CuNO}_3
ightarrow \text{Fe(NO}_3\text{)}_2 + \text{Cu}} \)
Step 1: Balance Fe
On the left, Fe has a coefficient of 1. On the right, \( \text{Fe(NO}_3\text{)}_2 \) has 1 Fe. So Fe is balanced initially (but we'll adjust later if needed).
Step 2: Balance \( \text{NO}_3 \)
On the right, \( \text{Fe(NO}_3\text{)}_2 \) has 2 \( \text{NO}_3 \) groups. So we need 2 \( \text{CuNO}_3 \) on the left to balance \( \text{NO}_3 \). Now the equation is \( \text{Fe} + 2\text{CuNO}_3
ightarrow \text{Fe(NO}_3\text{)}_2 + \text{Cu} \).
Step 3: Balance Cu
On the left, 2 \( \text{CuNO}_3 \) gives 2 Cu. So we need 2 Cu on the right. Now the equation is \( \text{Fe} + 2\text{CuNO}_3
ightarrow \text{Fe(NO}_3\text{)}_2 + 2\text{Cu} \).
Step 4: Check Fe (still balanced, 1 on each side)
- Fe: 1 (left) = 1 (right)
- Cu: 2 (left) = 2 (right)
- \( \text{NO}_3 \): 2 (left) = 2 (right)
Step 1: Balance Cl
On the left, NaCl has 1 Cl. On the right, \( \text{Cl}_2 \) has 2 Cl. So we need 2 NaCl on the left. Now the equation is \( 2\text{NaCl} + \text{F}_2
ightarrow \text{NaF} + \text{Cl}_2 \).
Step 2: Balance Na
On the left, 2 NaCl gives 2 Na. So we need 2 NaF on the right. Now the equation is \( 2\text{NaCl} + \text{F}_2
ightarrow 2\text{NaF} + \text{Cl}_2 \).
Step 3: Balance F
On the left, \( \text{F}_2 \) has 2 F. On the right, 2 NaF has 2 F. So F is balanced.
- Na: 2 (left) = 2 (right)
- Cl: 2 (left) = 2 (right)
- F: 2 (left) = 2 (right)
Step 1: Balance Al
On the left, Al has a coefficient of 1. On the right, \( \text{AlCl}_3 \) has 2 Al. So we need 2 Al on the left. Now the equation is \( 2\text{Al} + \text{CuCl}_2
ightarrow \text{Cu} + 2\text{AlCl}_3 \).
Step 2: Balance Cl
On the right, \( 2\text{AlCl}_3 \) has \( 2\times3 = 6 \) Cl. So we need 3 \( \text{CuCl}_2 \) on the left (since each \( \text{CuCl}_2 \) has 2 Cl, \( 3\times2 = 6 \) Cl). Now the equation is \( 2\text{Al} + 3\text{CuCl}_2
ightarrow \text{Cu} + 2\text{AlCl}_3 \).
Step 3: Balance Cu
On the left, 3 \( \text{CuCl}_2 \) gives 3 Cu. So we need 3 Cu on the right. Now the correct balanced equation is \( 2\text{Al} + 3\text{CuCl}_2
ightarrow 3\text{Cu} + 2\text{AlCl}_3 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1, 2, 1, 2 (coefficients for Fe, \( \text{CuNO}_3 \), \( \text{Fe(NO}_3\text{)}_2 \), Cu)
Problem 8: Balancing \( \boldsymbol{\text{NaCl} + \text{F}_2
ightarrow \text{NaF} + \text{Cl}_2} \)