QUESTION IMAGE
Question
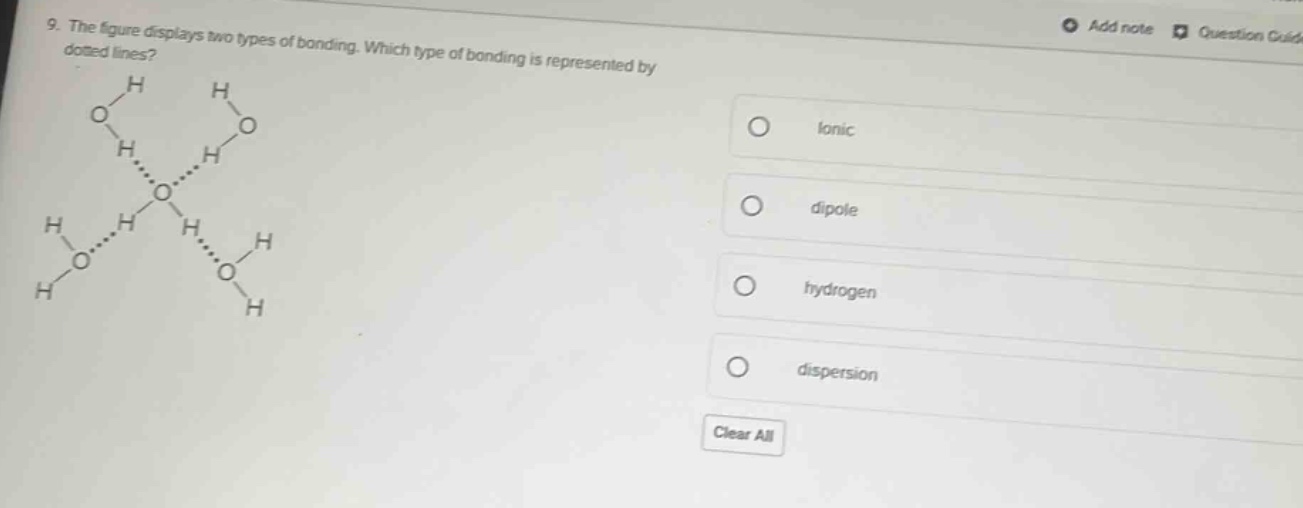
- the figure displays two types of bonding. which type of bonding is represented by dotted lines?
options:
- ionic
- dipole
- hydrogen
- dispersion
The figure shows water molecules (H₂O) with dotted lines between the oxygen of one molecule and the hydrogen of another. Hydrogen bonding occurs between a hydrogen atom bonded to a highly electronegative atom (like O in H₂O) and another electronegative atom (O, N, F) in a neighboring molecule. Ionic bonding involves transfer of electrons (not here), dipole - dipole is a general term for polar molecule interactions (but hydrogen bonding is a specific type of dipole - dipole for H with O/N/F), and dispersion is for non - polar molecules (water is polar). So the dotted lines represent hydrogen bonding.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. hydrogen