QUESTION IMAGE
Question
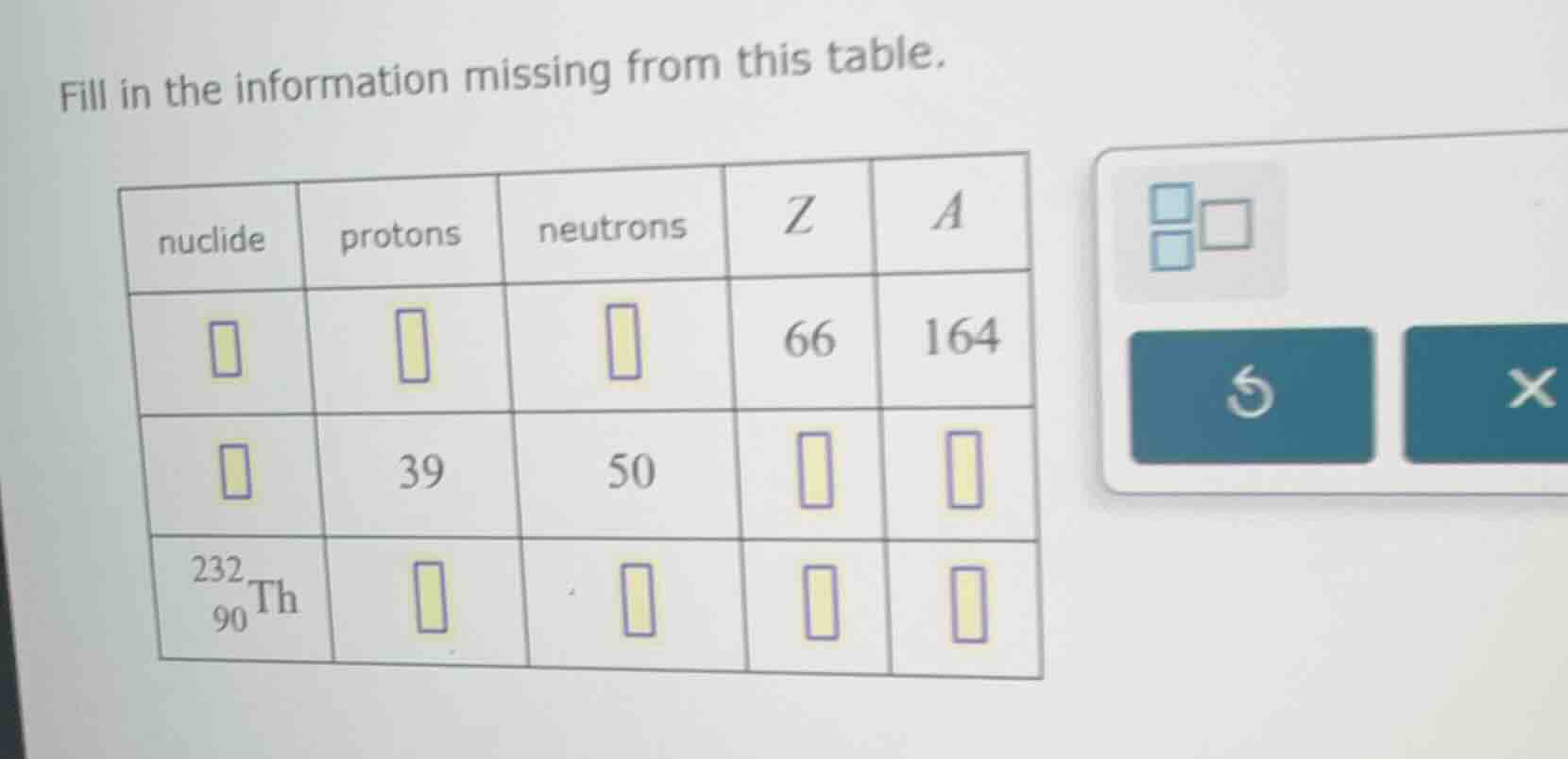
fill in the information missing from this table.
| nuclide | protons | neutrons | z | a |
|---|---|---|---|---|
| 39 | 50 | |||
| $_{90}^{232}\text{th}$ |
Step1: Recall definitions (Z=protons, A=protons+neutrons, nuclide symbol: element with Z, A)
For a nuclide, \( Z \) (atomic number) = number of protons. \( A \) (mass number) = number of protons + number of neutrons. The nuclide symbol is \( _Z^A X \), where \( X \) is the element with atomic number \( Z \).
Row 1: \( Z = 66 \), \( A = 164 \)
- Protons = \( Z = 66 \)
- Neutrons = \( A - Z = 164 - 66 = 98 \)
- Element with \( Z = 66 \) is Dysprosium (Dy), so nuclide: \( _{66}^{164}\text{Dy} \)
Row 2: Protons = 39, Neutrons = 50
- \( Z = \) protons = 39 (Element: Yttrium, Y)
- \( A = \) protons + neutrons = \( 39 + 50 = 89 \)
- Nuclide: \( _{39}^{89}\text{Y} \)
Row 3: \( _{90}^{232}\text{Th} \)
- Protons = \( Z = 90 \)
- Neutrons = \( A - Z = 232 - 90 = 142 \)
- \( Z = 90 \), \( A = 232 \)
Filled Table (Row-wise):
| nuclide | protons | neutrons | \( Z \) | \( A \) |
|---|---|---|---|---|
| \( _{39}^{89}\text{Y} \) | 39 | 50 | 39 | 89 |
| \( _{90}^{232}\text{Th} \) | 90 | 142 | 90 | 232 |
(Note: If you need individual cell answers:
Row 1: nuclide \( _{66}^{164}\text{Dy} \), protons 66, neutrons 98;
Row 2: nuclide \( _{39}^{89}\text{Y} \), \( Z = 39 \), \( A = 89 \);
Row 3: protons 90, neutrons 142, \( Z = 90 \), \( A = 232 \))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Step1: Recall definitions (Z=protons, A=protons+neutrons, nuclide symbol: element with Z, A)
For a nuclide, \( Z \) (atomic number) = number of protons. \( A \) (mass number) = number of protons + number of neutrons. The nuclide symbol is \( _Z^A X \), where \( X \) is the element with atomic number \( Z \).
Row 1: \( Z = 66 \), \( A = 164 \)
- Protons = \( Z = 66 \)
- Neutrons = \( A - Z = 164 - 66 = 98 \)
- Element with \( Z = 66 \) is Dysprosium (Dy), so nuclide: \( _{66}^{164}\text{Dy} \)
Row 2: Protons = 39, Neutrons = 50
- \( Z = \) protons = 39 (Element: Yttrium, Y)
- \( A = \) protons + neutrons = \( 39 + 50 = 89 \)
- Nuclide: \( _{39}^{89}\text{Y} \)
Row 3: \( _{90}^{232}\text{Th} \)
- Protons = \( Z = 90 \)
- Neutrons = \( A - Z = 232 - 90 = 142 \)
- \( Z = 90 \), \( A = 232 \)
Filled Table (Row-wise):
| nuclide | protons | neutrons | \( Z \) | \( A \) |
|---|---|---|---|---|
| \( _{39}^{89}\text{Y} \) | 39 | 50 | 39 | 89 |
| \( _{90}^{232}\text{Th} \) | 90 | 142 | 90 | 232 |
(Note: If you need individual cell answers:
Row 1: nuclide \( _{66}^{164}\text{Dy} \), protons 66, neutrons 98;
Row 2: nuclide \( _{39}^{89}\text{Y} \), \( Z = 39 \), \( A = 89 \);
Row 3: protons 90, neutrons 142, \( Z = 90 \), \( A = 232 \))