QUESTION IMAGE
Question
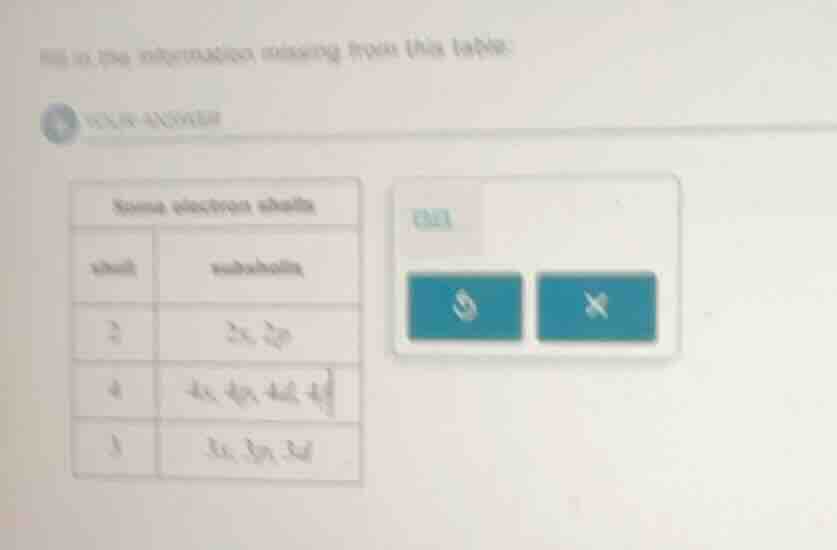
fill in the information missing from this table: some electron shells shell subshells 2 2s, 2p 4 4s, 4p, 4d, 4f 3 3s, 3p, 3d
To fill in the missing subshell for shell 2, we recall the electron subshells for each principal quantum number \( n \) (shell number). For \( n = 2 \), the possible subshells (azimuthal quantum number \( l \)) are \( l = 0 \) (s - subshell) and \( l = 1 \) (p - subshell), but also, we need to check the correct subshells. Wait, actually, for shell \( n = 2 \), the subshells are \( 2s \) and \( 2p \), but wait, maybe there's a missing one? Wait, no, the principal quantum number \( n \) determines the possible \( l \) values as \( l = 0, 1, ..., n - 1 \). So for \( n = 2 \), \( l = 0 \) (s) and \( l = 1 \) (p). But the table has "2s, 2p" but maybe a typo? Wait, no, the user is to fill the missing. Wait, the shell 1: what's shell 1? Wait, the table has shell 2, 4, 3? Wait, no, the original table:
Wait, the table has:
- Shell 2: subshells 2s, 2p (but maybe missing 2d? No, \( n = 2 \), \( l \) can be 0,1. \( l = 2 \) would be d, but \( n = 2 \), \( l \) max is 1 (since \( l < n \)), so 2d doesn't exist. Wait, maybe the shell 1? Wait, the first row: "shell" and "subshells". Then:
- Shell 2: 2s, 2p (maybe missing 2d? No, \( n=2 \), \( l \) can be 0,1. So 2s (l=0), 2p (l=1). So maybe the shell 1? Wait, the user's table:
Looking at the table, the rows are:
- Shell 2: 2s, 2p (maybe missing 2d? No, \( n=2 \), \( l \) can't be 2. So maybe the shell 1? Wait, the shell 1: \( n=1 \), \( l=0 \), so subshell 1s.
Wait, the problem says "fill in the information missing from this table". Let's check the shells:
- Shell 1: subshell 1s (since \( n=1 \), \( l=0 \))
- Shell 2: subshells 2s, 2p (since \( n=2 \), \( l=0,1 \))
- Shell 3: subshells 3s, 3p, 3d (since \( n=3 \), \( l=0,1,2 \))
- Shell 4: subshells 4s, 4p, 4d, 4f (since \( n=4 \), \( l=0,1,2,3 \))
Wait, the table has:
- Shell 2: 2s, 2p (maybe missing 2d? No, \( n=2 \), \( l \) max 1. So no. Wait, maybe the shell 1 is missing? Let's see the original image: the table has rows for shell 2, 4, 3? Wait, the user's table:
Row 1: shell 2, subshells 2s, 2p
Row 2: shell 4, subshells 4s, 4p, 4d, 4f
Row 3: shell 3, subshells 3s, 3p, 3d
Wait, maybe the first row is shell 1? Let's check:
Shell 1: \( n=1 \), \( l=0 \), so subshell 1s.
So the missing shell is shell 1, with subshell 1s.
Wait, the user's input: "fill in the information missing from this table". The table has shell 2, 4, 3. So the missing shell is shell 1, subshell 1s.
So the missing part is shell 1 with subshell 1s.
Step1: Recall electron subshell rules
For a shell with principal quantum number \( n \), subshells (azimuthal quantum number \( l \)) satisfy \( l = 0, 1, ..., n - 1 \).
Step2: Analyze shell 1 (\( n = 1 \))
For \( n = 1 \), \( l \) can only be \( 0 \) (since \( l < n \)). The subshell for \( l = 0 \) is \( s \), so the subshell is \( 1s \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Shell 1: subshell \( 1s \) (so the missing row is shell 1 with subshell \( 1s \))