QUESTION IMAGE
Question
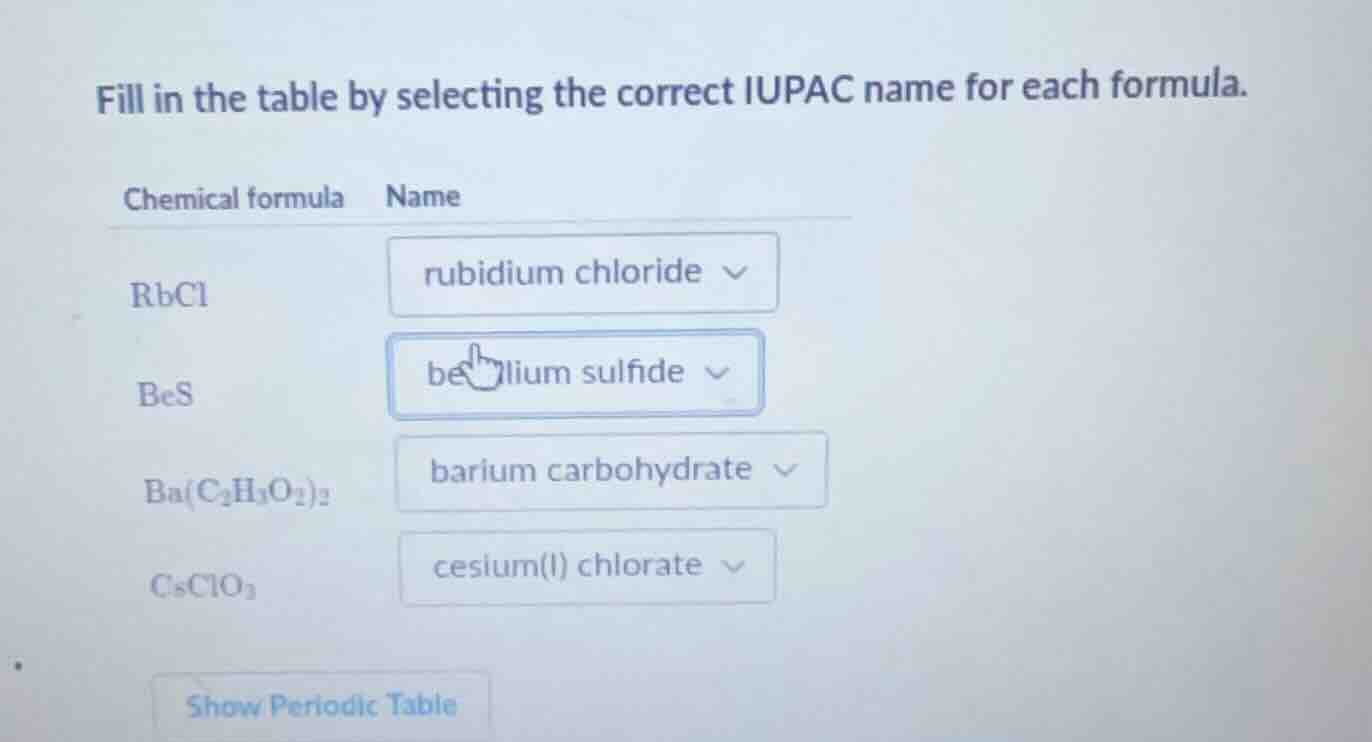
fill in the table by selecting the correct iupac name for each formula.
chemical formula name
rbcl rubidium chloride
bes beryllium sulfide
ba(c₂h₃o₂)₂ barium carbohydrate
csclo₃ cesium(i) chlorate
show periodic table
Response
To solve this, we analyze each chemical formula and correct its IUPAC name:
For \(\boldsymbol{\ce{RbCl}}\):
- \( \ce{Rb} \) is rubidium (not "rubidium" misspelled as "rubidium"—wait, the original has "rubidium" as "rubidium"? No, the error is the spelling: "rubidium" (correct) vs. "rubidium" (wait, no, the original dropdown has "rubidium chloride" (correct spelling: "rubidium" is correct, "rubidium" is the element). Wait, no—\( \ce{Rb} \) is rubidium, so "rubidium chloride" is correct. Wait, maybe the user’s image has a typo, but assuming the formula is \( \ce{RbCl} \), the name is rubidium chloride (correct spelling: "rubidium" not "rubidium"—wait, "rubidium" is correct. So if the dropdown has "rubidium chloride", that’s correct.
For \(\boldsymbol{\ce{BeS}}\):
- \( \ce{Be} \) is beryllium, \( \ce{S} \) is sulfide (sulfide ion \( \ce{S^{2-}} \)).
- Correct name: beryllium sulfide (the dropdown has "beryllium sulfide"—wait, the user’s image shows "beryllium sulfide" as the option, which is correct.
For \(\boldsymbol{\ce{Ba(C2H3O2)2}}\):
- \( \ce{C2H3O2^-} \) is the acetate ion (\( \ce{CH3COO^-} \), acetate).
- \( \ce{Ba} \) is barium (charge \( +2 \), so 2 acetate ions balance it).
- Correct name: barium acetate (not "barium carbohydrate"—carbohydrate is organic, not an ion. Acetate is the anion here).
For \(\boldsymbol{\ce{CsClO3}}\):
- \( \ce{Cs} \) is cesium (group 1, charge \( +1 \), so no need to specify (I) in the name, as cesium only forms \( +1 \) ions).
- \( \ce{ClO3^-} \) is chlorate ion.
- Correct name: cesium chlorate (not "cesium(I) chlorate"—cesium’s charge is always \( +1 \), so the (I) is unnecessary).
Final Table (Corrected):
| Chemical Formula | Correct IUPAC Name |
|---|---|
| \( \ce{BeS} \) | beryllium sulfide |
| \( \ce{Ba(C2H3O2)2} \) | barium acetate |
| \( \ce{CsClO3} \) | cesium chlorate |
Summary of Corrections:
- \( \ce{Ba(C2H3O2)2} \): Change from "barium carbohydrate" to "barium acetate".
- \( \ce{CsClO3} \): Change from "cesium(I) chlorate" to "cesium chlorate".
- \( \ce{BeS} \): "beryllium sulfide" is correct (assuming the dropdown’s "beryllium" was a typo, but if the option is "beryllium sulfide", that’s correct).
- \( \ce{RbCl} \): "rubidium chloride" is correct (spelling: "rubidium" is correct).
Final Answers (Table):
| Chemical Formula | Name |
|---|---|
| \( \ce{BeS} \) | beryllium sulfide |
| \( \ce{Ba(C2H3O2)2} \) | barium acetate |
| \( \ce{CsClO3} \) | cesium chlorate |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each chemical formula and correct its IUPAC name:
For \(\boldsymbol{\ce{RbCl}}\):
- \( \ce{Rb} \) is rubidium (not "rubidium" misspelled as "rubidium"—wait, the original has "rubidium" as "rubidium"? No, the error is the spelling: "rubidium" (correct) vs. "rubidium" (wait, no, the original dropdown has "rubidium chloride" (correct spelling: "rubidium" is correct, "rubidium" is the element). Wait, no—\( \ce{Rb} \) is rubidium, so "rubidium chloride" is correct. Wait, maybe the user’s image has a typo, but assuming the formula is \( \ce{RbCl} \), the name is rubidium chloride (correct spelling: "rubidium" not "rubidium"—wait, "rubidium" is correct. So if the dropdown has "rubidium chloride", that’s correct.
For \(\boldsymbol{\ce{BeS}}\):
- \( \ce{Be} \) is beryllium, \( \ce{S} \) is sulfide (sulfide ion \( \ce{S^{2-}} \)).
- Correct name: beryllium sulfide (the dropdown has "beryllium sulfide"—wait, the user’s image shows "beryllium sulfide" as the option, which is correct.
For \(\boldsymbol{\ce{Ba(C2H3O2)2}}\):
- \( \ce{C2H3O2^-} \) is the acetate ion (\( \ce{CH3COO^-} \), acetate).
- \( \ce{Ba} \) is barium (charge \( +2 \), so 2 acetate ions balance it).
- Correct name: barium acetate (not "barium carbohydrate"—carbohydrate is organic, not an ion. Acetate is the anion here).
For \(\boldsymbol{\ce{CsClO3}}\):
- \( \ce{Cs} \) is cesium (group 1, charge \( +1 \), so no need to specify (I) in the name, as cesium only forms \( +1 \) ions).
- \( \ce{ClO3^-} \) is chlorate ion.
- Correct name: cesium chlorate (not "cesium(I) chlorate"—cesium’s charge is always \( +1 \), so the (I) is unnecessary).
Final Table (Corrected):
| Chemical Formula | Correct IUPAC Name |
|---|---|
| \( \ce{BeS} \) | beryllium sulfide |
| \( \ce{Ba(C2H3O2)2} \) | barium acetate |
| \( \ce{CsClO3} \) | cesium chlorate |
Summary of Corrections:
- \( \ce{Ba(C2H3O2)2} \): Change from "barium carbohydrate" to "barium acetate".
- \( \ce{CsClO3} \): Change from "cesium(I) chlorate" to "cesium chlorate".
- \( \ce{BeS} \): "beryllium sulfide" is correct (assuming the dropdown’s "beryllium" was a typo, but if the option is "beryllium sulfide", that’s correct).
- \( \ce{RbCl} \): "rubidium chloride" is correct (spelling: "rubidium" is correct).
Final Answers (Table):
| Chemical Formula | Name |
|---|---|
| \( \ce{BeS} \) | beryllium sulfide |
| \( \ce{Ba(C2H3O2)2} \) | barium acetate |
| \( \ce{CsClO3} \) | cesium chlorate |