QUESTION IMAGE
Question
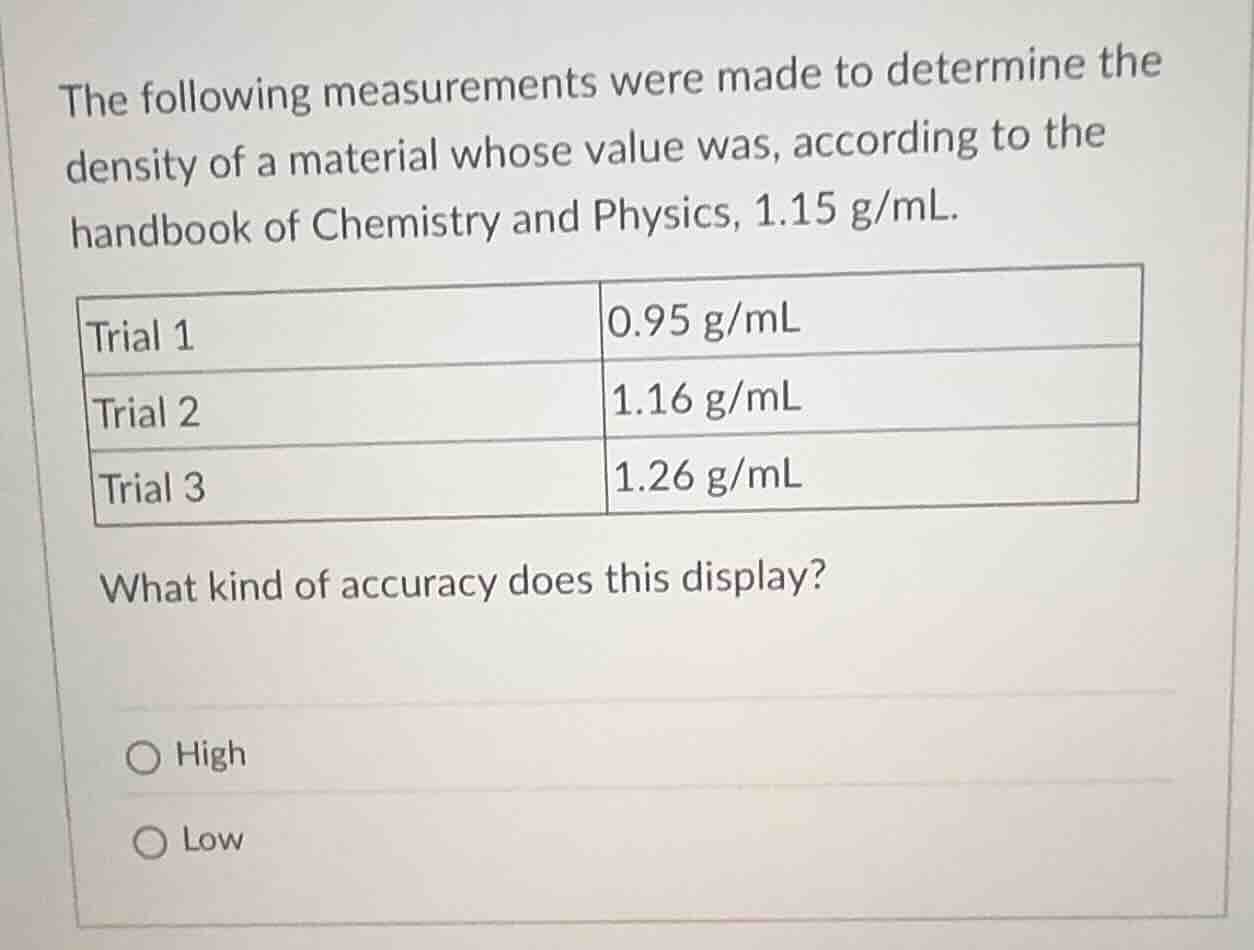
the following measurements were made to determine the density of a material whose value was, according to the handbook of chemistry and physics, 1.15 g/ml.
| trial 1 | 0.95 g/ml |
| trial 2 | 1.16 g/ml |
| trial 3 | 1.26 g/ml |
what kind of accuracy does this display?
- high
- low
Brief Explanations
Accuracy refers to how close measured values are to the true value (1.15 g/mL here). Trial 1 (0.95) is far from 1.15, Trial 2 (1.16) is close, but Trial 3 (1.26) is also off. Since not all trials are close to the true value, and one is significantly different, the overall accuracy is low.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Low