QUESTION IMAGE
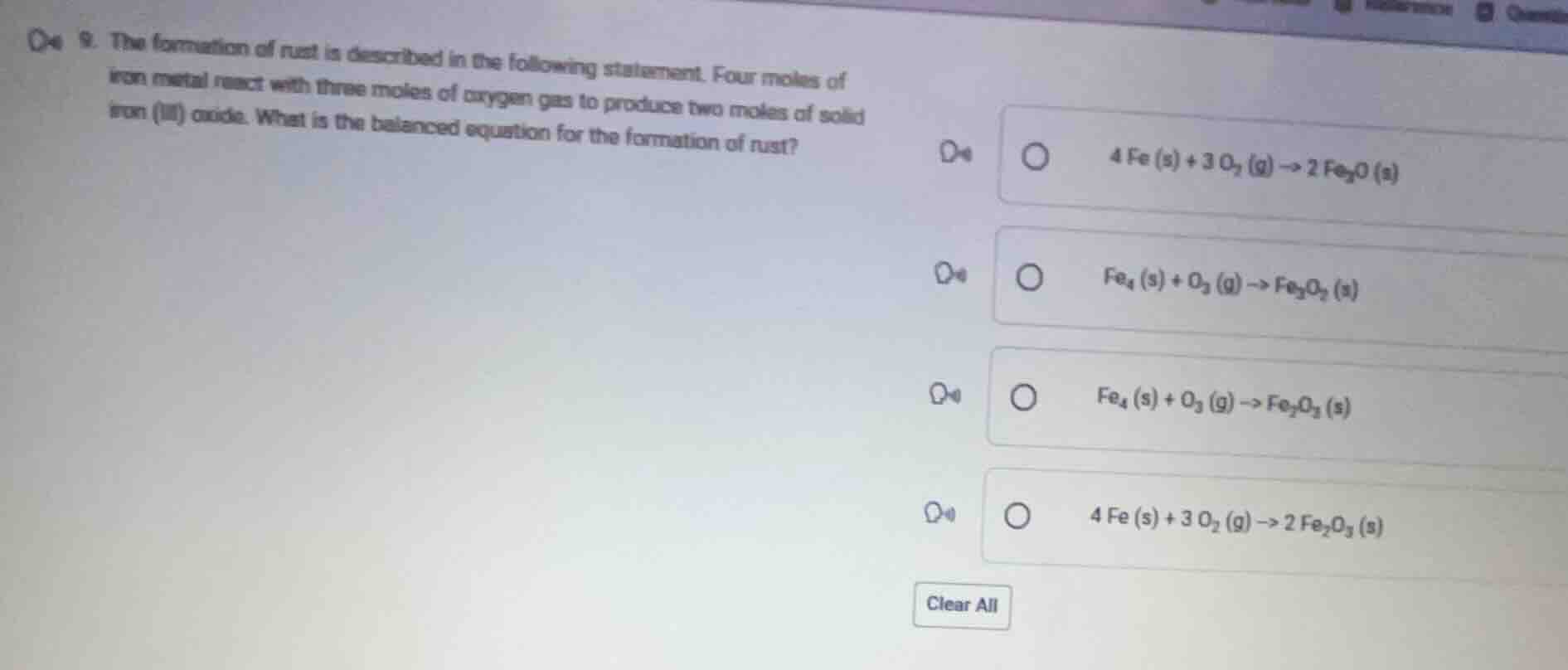
Question
- the formation of rust is described in the following statement. four moles of iron metal react with three moles of oxygen gas to produce two moles of solid iron (iii) oxide. what is the balanced equation for the formation of rust? \\( 4 fe (s) + 3 o_2 (g) \
ightarrow 2 fe_3o (s) \\) \\( fe_4 (s) + o_3 (g) \
ightarrow fe_3o_2 (s) \\) \\( fe_4 (s) + o_3 (g) \
ightarrow fe_2o_3 (s) \\) \\( 4 fe (s) + 3 o_2 (g) \
ightarrow 2 fe_2o_3 (s) \\)
Brief Explanations
- First, identify the reactants and products: Reactants are iron (Fe) and oxygen gas ($O_2$), product is iron(III) oxide ($Fe_2O_3$).
- The problem states 4 moles of Fe, 3 moles of $O_2$, and 2 moles of $Fe_2O_3$ are involved.
- Check each option:
- Option 1: Product is $Fe_3O$ which is incorrect (iron(III) oxide is $Fe_2O_3$).
- Option 2: Reactants are $Fe_4$ and $O_3$ which don't match the problem's reactants (Fe and $O_2$).
- Option 3: Same as option 2, incorrect reactants.
- Option 4: Reactants are 4 Fe and 3 $O_2$, product is 2 $Fe_2O_3$. Let's check atoms:
- Fe: 4 on left, 2×2 = 4 on right (balanced).
- O: 3×2 = 6 on left, 2×3 = 6 on right (balanced). This matches the problem's description.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $4 Fe (s) + 3 O_2 (g)
ightarrow 2 Fe_2O_3 (s)$