QUESTION IMAGE
Question
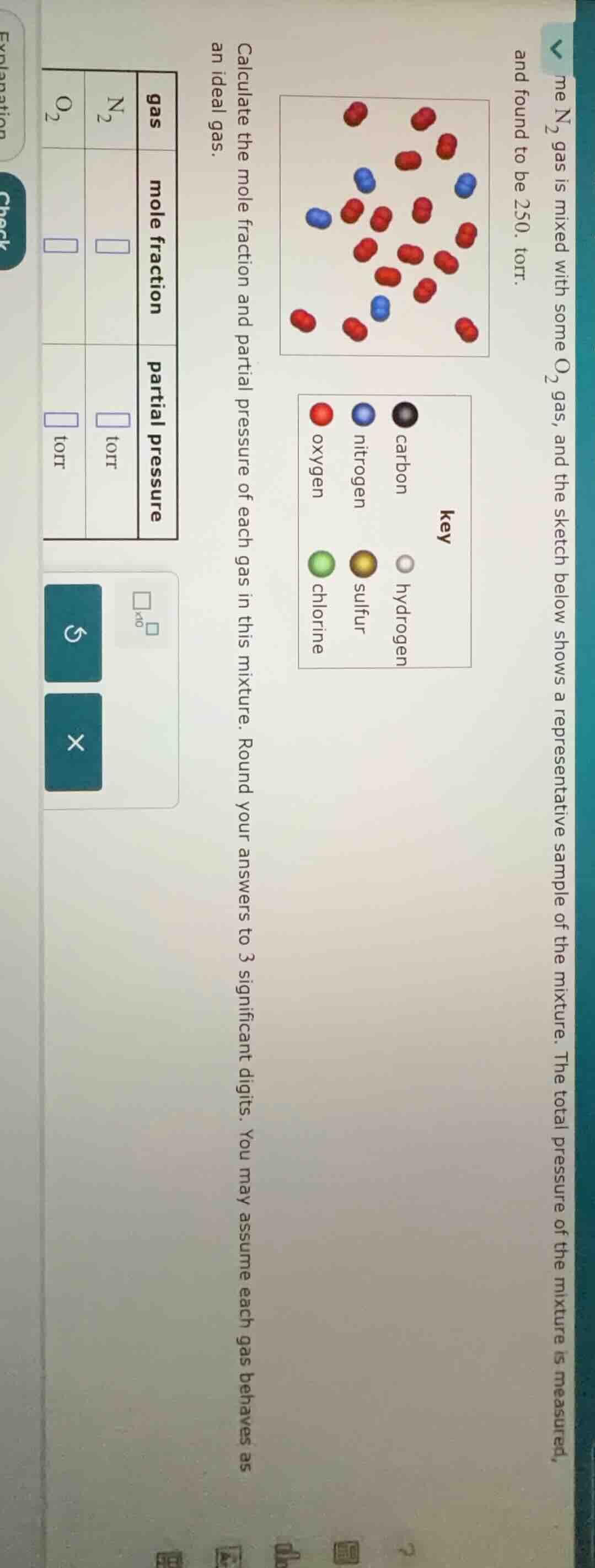
me n₂ gas is mixed with some o₂ gas, and the sketch below shows a representative sample of the mixture. the total pressure of the mixture is measured, and found to be 250. torr. calculate the mole fraction and partial pressure of each gas in this mixture. round your answers to 3 significant digits. you may assume each gas behaves as an ideal gas.
Step1: Count the number of molecules
From the sketch, count the number of \(N_2\) (blue) and \(O_2\) (red) molecules. Let's say there are \(n_{N_2} = 4\) (blue molecules) and \(n_{O_2}=14\) (red molecules). The total number of moles (since each molecule is a mole for ideal gas approximation) \(n_{total}=n_{N_2} + n_{O_2}=4 + 14 = 18\).
Step2: Calculate mole fraction of \(N_2\)
Mole fraction of \(N_2\), \(X_{N_2}=\frac{n_{N_2}}{n_{total}}=\frac{4}{18}\approx0.222\)
Step3: Calculate mole fraction of \(O_2\)
Mole fraction of \(O_2\), \(X_{O_2}=\frac{n_{O_2}}{n_{total}}=\frac{14}{18}\approx0.778\)
Step4: Calculate partial pressure of \(N_2\)
Total pressure \(P_{total} = 250\) torr. Partial pressure of \(N_2\), \(P_{N_2}=X_{N_2}\times P_{total}=0.222\times250 = 55.5\) torr
Step5: Calculate partial pressure of \(O_2\)
Partial pressure of \(O_2\), \(P_{O_2}=X_{O_2}\times P_{total}=0.778\times250 = 194.5\approx195\) torr (rounded to 3 significant digits)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| gas | mole fraction | partial pressure (torr) |
|---|---|---|
| \(O_2\) | \(0.778\) | \(195\) |