QUESTION IMAGE
Question
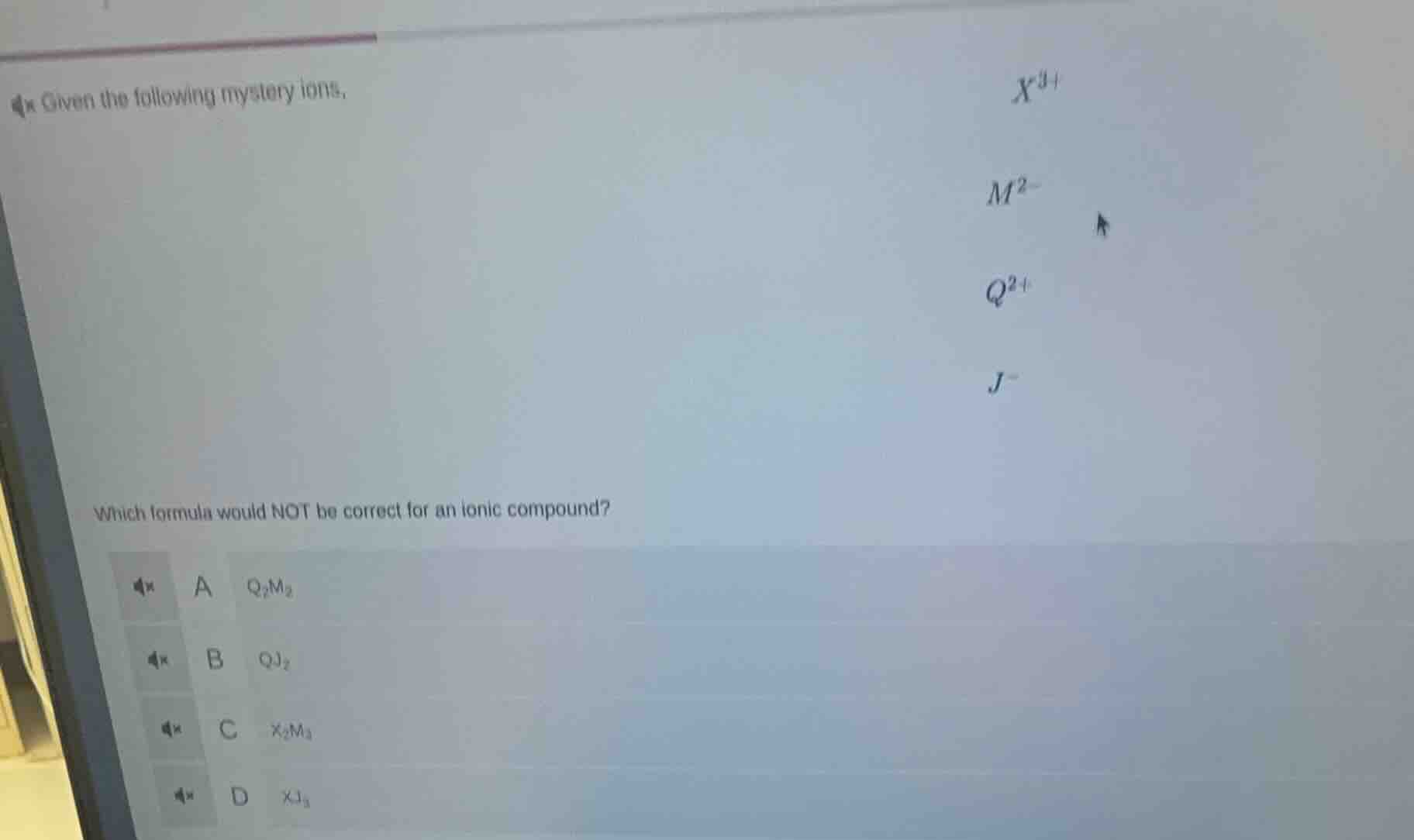
given the following mystery ions, \\(x^{3+}\\) \\(m^{2-}\\) \\(q^{2+}\\) \\(j^-\\) which formula would not be correct for an ionic compound? a \\(q_2m_2\\) b \\(qj_2\\) c \\(x_2m_3\\) d \\(xj_3\\)
To determine the correct ionic compound formula, we use the criss - cross method, where the charge of one ion becomes the subscript of the other.
Step 1: Analyze Option A ($Q_2M_2$)
- The ion $Q$ has a charge of $2 +$ ($Q^{2+}$) and the ion $M$ has a charge of $2 -$ ($M^{2 -}$).
- Using the criss - cross method, the formula should be $Q_2M_2$? Wait, no. Let's do it properly. The ratio of $Q^{2+}$ to $M^{2 -}$: the magnitude of the charges are equal (2 and 2). So the formula should be $QM$ (since the subscripts of 2 can be simplified by dividing by 2). So $Q_2M_2$ is not the simplest form and is incorrect. But let's check other options to be sure.
Step 2: Analyze Option B ($QJ_2$)
- $Q$ is $Q^{2+}$ and $J$ is $J^-$.
- Using criss - cross: the charge of $Q$ (2) becomes the subscript of $J$, and the charge of $J$ (1) becomes the subscript of $Q$. So the formula is $Q_1J_2$ or $QJ_2$. This is correct.
Step 3: Analyze Option C ($X_2M_3$)
- $X$ is $X^{3+}$ and $M$ is $M^{2 -}$.
- Criss - cross: charge of $X$ (3) becomes subscript of $M$, charge of $M$ (2) becomes subscript of $X$. So the formula is $X_2M_3$. This is correct.
Step 4: Analyze Option D ($XJ_3$)
- $X$ is $X^{3+}$ and $J$ is $J^-$.
- Criss - cross: charge of $X$ (3) becomes subscript of $J$, charge of $J$ (1) becomes subscript of $X$. So the formula is $X_1J_3$ or $XJ_3$. This is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $Q_2M_2$