QUESTION IMAGE
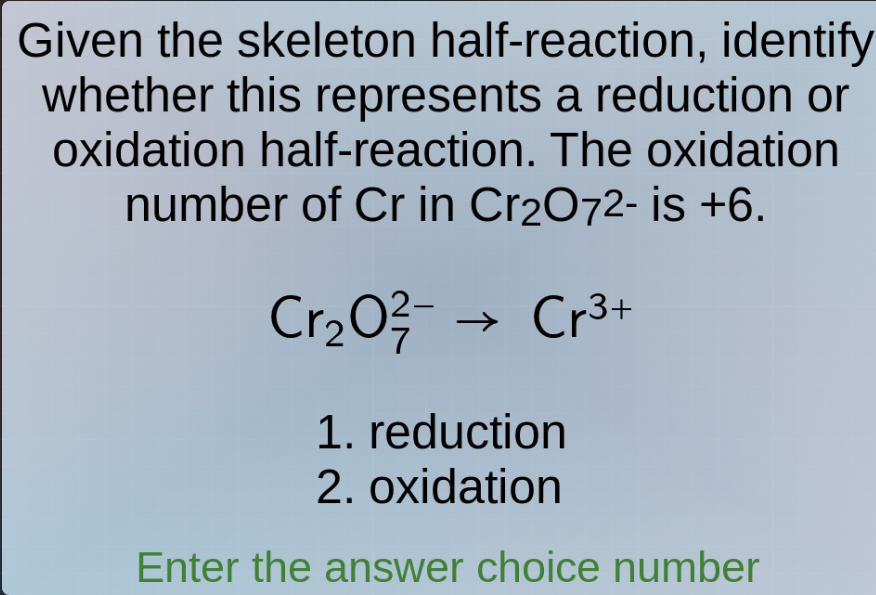
Question
given the skeleton half-reaction, identify whether this represents a reduction or oxidation half-reaction. the oxidation number of cr in cr₂o₇²⁻ is +6. cr₂o₇²⁻ → cr³⁺ 1. reduction 2. oxidation enter the answer choice number
To determine if it's reduction or oxidation, we check the change in oxidation number of Cr. In $\ce{Cr2O7^{2-}}$, Cr has an oxidation number of +6. In $\ce{Cr^{3+}}$, the oxidation number is +3. A decrease in oxidation number (from +6 to +3) indicates a gain of electrons, which is reduction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- reduction