QUESTION IMAGE
Question
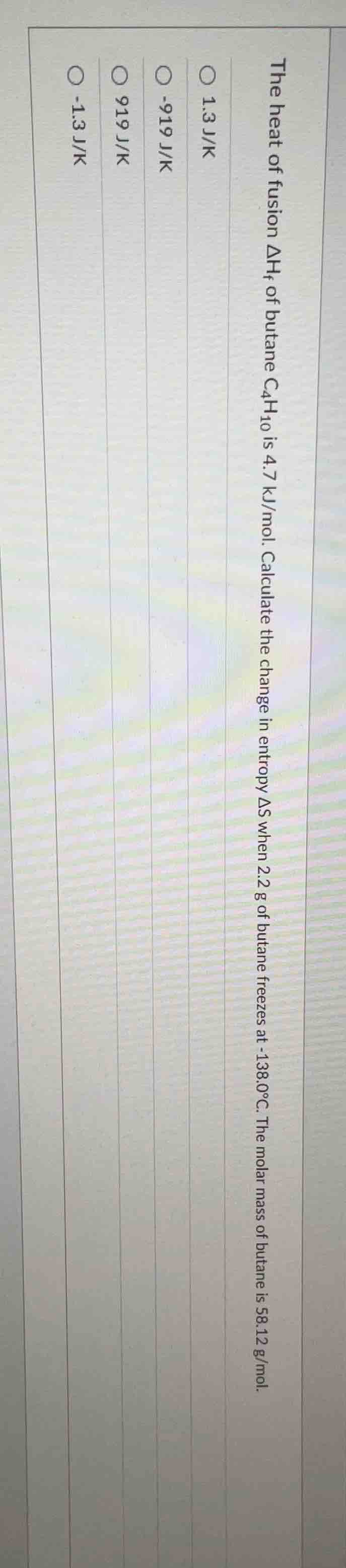
the heat of fusion $\delta h_f$ of butane $c_4h_{10}$ is 4.7 kj/mol. calculate the change in entropy $\delta s$ when 2.2 g of butane freezes at -138.0$^\circ$c. the molar mass of butane is 58.12 g/mol. \bigcirc 1.3 j/k \bigcirc -919 j/k \bigcirc 919 j/k \bigcirc -1.3 j/k
Step1: Calculate moles of butane
Moles ($n$) = $\frac{\text{mass}}{\text{molar mass}}$. Mass = 2.2 g, molar mass = 58.12 g/mol.
$n = \frac{2.2}{58.12} \approx 0.03785$ mol.
Step2: Determine $\Delta H$ for freezing
Fusion is melting, freezing is reverse. So $\Delta H_{\text{freezing}} = -\Delta H_{\text{fusion}} = -4.7$ kJ/mol = -4700 J/mol.
Step3: Calculate heat change ($q$)
$q = n \times \Delta H_{\text{freezing}} = 0.03785 \text{ mol} \times (-4700 \text{ J/mol}) \approx -177.895$ J.
Step4: Convert temperature to Kelvin
$T = -138.0^\circ\text{C} + 273.15 = 135.15$ K.
Step5: Calculate entropy change ($\Delta S$)
For phase change, $\Delta S = \frac{q}{T}$ (at constant $T$).
$\Delta S = \frac{-177.895 \text{ J}}{135.15 \text{ K}} \approx -1.316$ J/K $\approx -1.3$ J/K (or check options, closest is -1.3 J/K? Wait, wait, maybe miscalculation. Wait, wait, fusion $\Delta H_f$ is 4.7 kJ/mol, so freezing is -4.7 kJ/mol. Wait, maybe I messed moles. Wait 2.2g /58.12g/mol = 0.03785 mol. Then $q = 0.03785 \times (-4700) = -177.895$ J. $T = -138 + 273.15 = 135.15$ K. $\Delta S = q/T = -177.895 /135.15 ≈ -1.316 ≈ -1.3$ J/K. Wait but let's recheck. Wait, maybe the question is about fusion (melting) but freezing is opposite. Wait, entropy change for freezing: since it's a phase transition, $\Delta S = \frac{\Delta H}{T}$, but $\Delta H$ for freezing is $-\Delta H_{fusion}$. So $\Delta S = \frac{-\Delta H_{fusion} \times n}{T}$. Wait, no: $\Delta S = \frac{q_{rev}}{T}$, and $q_{rev}$ for freezing is $n \times (-\Delta H_{fusion})$. So yes, as above. Wait, but let's recalculate moles: 2.2 /58.12 = 0.03785 mol. $\Delta H_{fusion} = 4700$ J/mol, so $\Delta H_{freezing} = -4700$ J/mol. $q = 0.03785 \times (-4700) = -177.895$ J. $T = 135.15$ K. $\Delta S = -177.895 /135.15 ≈ -1.316 ≈ -1.3$ J/K. So the option -1.3 J/K. Wait, but let's check again. Wait, maybe I made a mistake in temperature? -138.0 +273.15 = 135.15 K. Correct. Moles: 2.2/58.12 ≈0.03785. Correct. So $\Delta S ≈ -1.3$ J/K. So the answer is -1.3 J/K. Wait, but let's check the options: the last option is -1.3 J/K. So that's the answer.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
-1.3 J/K