QUESTION IMAGE
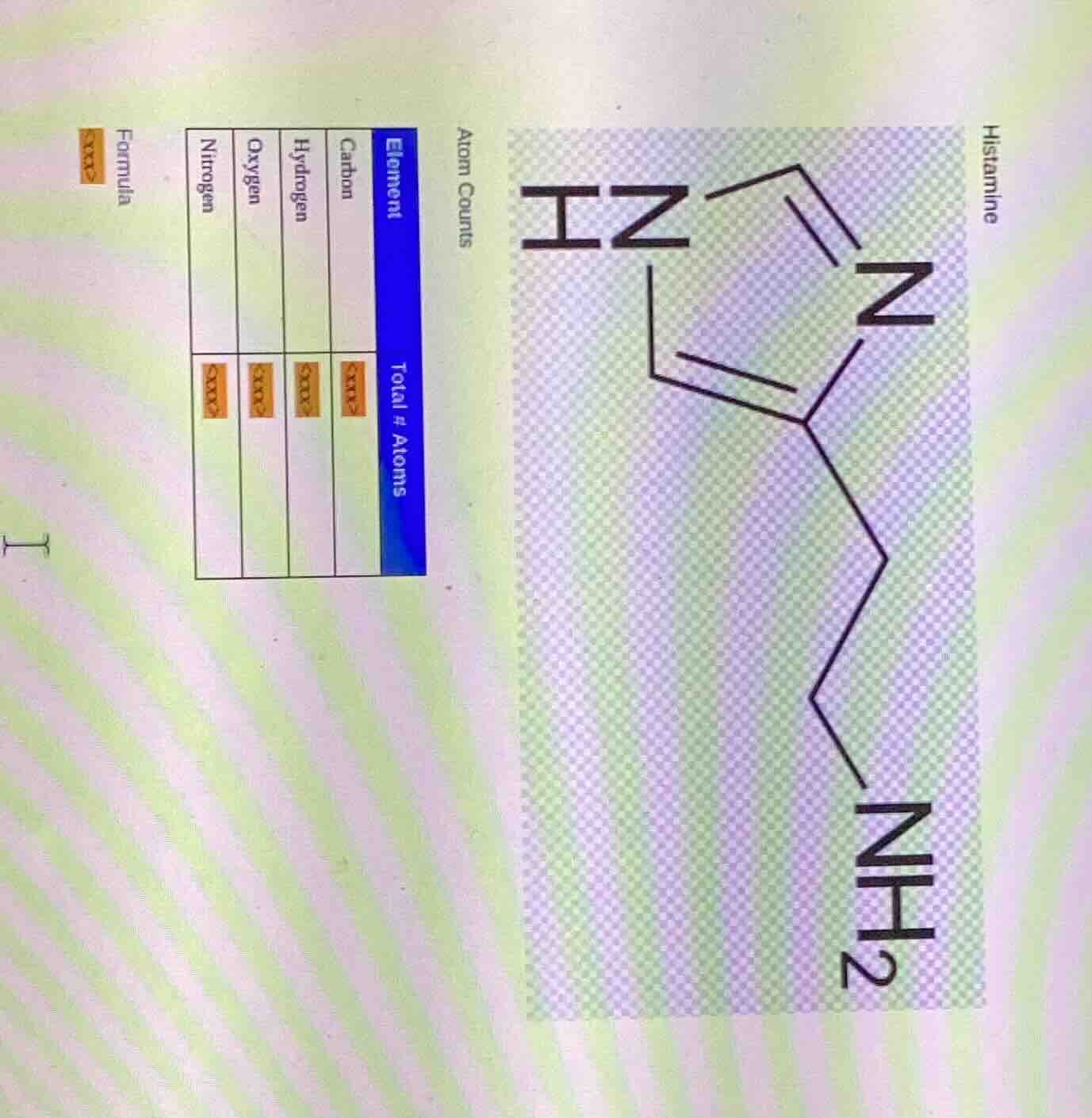
Question
histamine
atom counts
element\ttotal # atoms
carbon\t<xxx>
hydrogen\t<xxx>
oxygen\t<xxx>
nitrogen\t<xxx>
formula <xxx>
To determine the atom counts for histamine (\( \text{C}_5\text{H}_9\text{N}_3 \), since histamine's molecular formula is \( \text{C}_5\text{H}_9\text{N}_3 \) and there is no oxygen):
Step 1: Carbon Atoms
In the histamine structure, the carbon atoms in the imidazole ring (5 - membered ring with 2 N atoms) and the \( -\text{CH}_2-\text{CH}_2-\text{NH}_2 \) chain: total carbon atoms \( = 5 \).
Step 2: Hydrogen Atoms
Counting hydrogens: in the ring (each carbon in ring has 1 H, except where bonded to chain), and in \( -\text{NH}_2 \) (2 H) and the \( -\text{CH}_2-\text{CH}_2- \) (2 + 2 H). Total: \( 9 \).
Step 3: Nitrogen Atoms
Imidazole ring has 2 N, and \( -\text{NH}_2 \) has 1 N: total \( = 3 \).
Step 4: Oxygen Atoms
Histamine has no oxygen atoms, so count \( = 0 \).
| Element | Total # Atoms |
|---|---|
| Hydrogen | 9 |
| Oxygen | 0 |
| Nitrogen | 3 |
If the table was to be filled with these values, the cells for Carbon, Hydrogen, Oxygen, Nitrogen would be 5, 9, 0, 3 respectively.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine the atom counts for histamine (\( \text{C}_5\text{H}_9\text{N}_3 \), since histamine's molecular formula is \( \text{C}_5\text{H}_9\text{N}_3 \) and there is no oxygen):
Step 1: Carbon Atoms
In the histamine structure, the carbon atoms in the imidazole ring (5 - membered ring with 2 N atoms) and the \( -\text{CH}_2-\text{CH}_2-\text{NH}_2 \) chain: total carbon atoms \( = 5 \).
Step 2: Hydrogen Atoms
Counting hydrogens: in the ring (each carbon in ring has 1 H, except where bonded to chain), and in \( -\text{NH}_2 \) (2 H) and the \( -\text{CH}_2-\text{CH}_2- \) (2 + 2 H). Total: \( 9 \).
Step 3: Nitrogen Atoms
Imidazole ring has 2 N, and \( -\text{NH}_2 \) has 1 N: total \( = 3 \).
Step 4: Oxygen Atoms
Histamine has no oxygen atoms, so count \( = 0 \).
| Element | Total # Atoms |
|---|---|
| Hydrogen | 9 |
| Oxygen | 0 |
| Nitrogen | 3 |
If the table was to be filled with these values, the cells for Carbon, Hydrogen, Oxygen, Nitrogen would be 5, 9, 0, 3 respectively.