QUESTION IMAGE
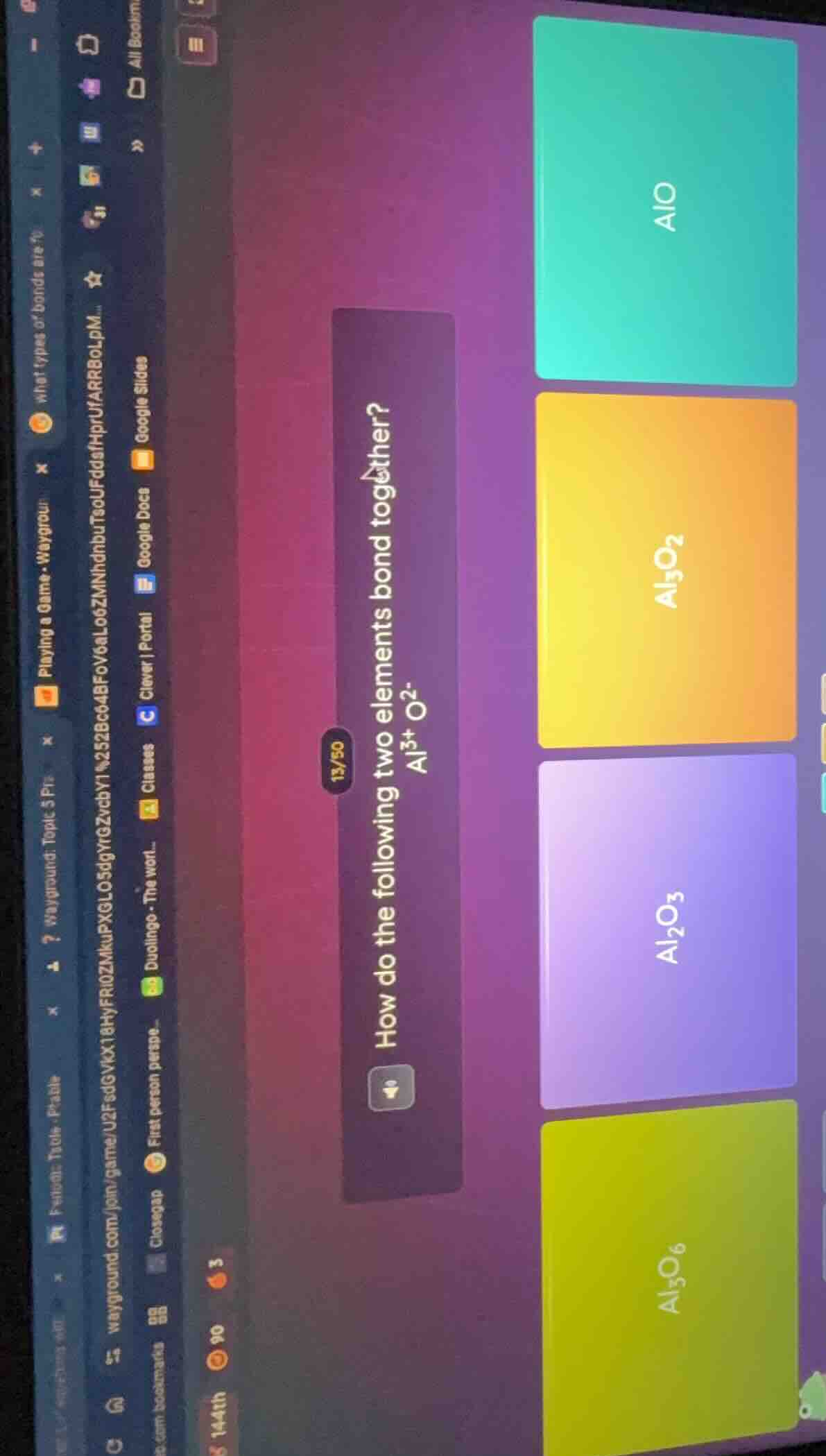
Question
how do the following two elements bond together? al³⁺ o²⁻ options: alo, al₃o₂, al₂o₃, al₃o₆
Step1: Balance cation/anion charges
Aluminum has a +3 charge ($\text{Al}^{3+}$), oxygen has a -2 charge ($\text{O}^{2-}$). To balance total charge to 0:
Let number of Al = $x$, number of O = $y$.
$3x - 2y = 0$
Step2: Find smallest integer values
Solve for $x$ and $y$:
$3x = 2y$
Smallest integers: $x=2$, $y=3$ (since $3*2=6$, $2*3=6$; charges cancel: $+6-6=0$)
Step3: Write neutral formula
Combine the ions with the found subscripts: $\text{Al}_2\text{O}_3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{Al}_2\text{O}_3$ (the purple option)