QUESTION IMAGE
Question
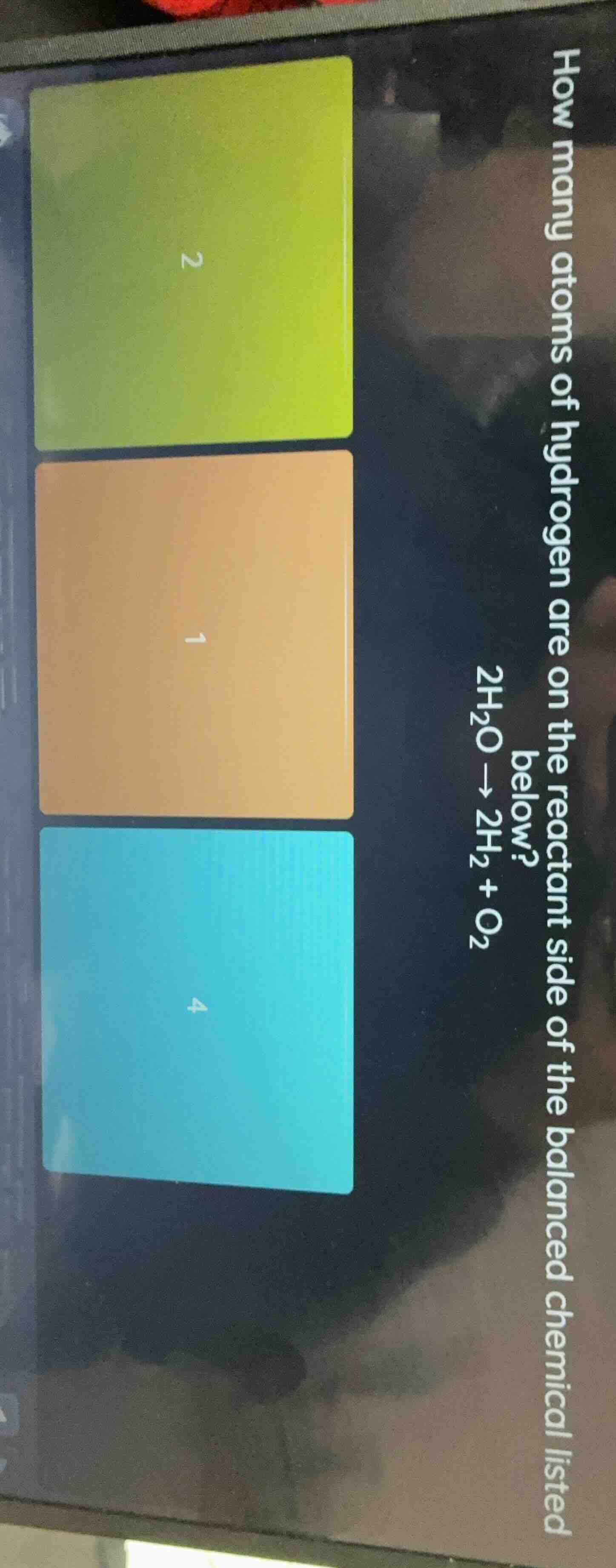
how many atoms of hydrogen are on the reactant side of the balanced chemical listed below? 2h₂o → 2h₂ + o₂
Step1: Analyze the reactant formula
The reactant is \(2H_2O\). The coefficient is 2, and in each \(H_2O\) molecule, there are 2 hydrogen atoms.
Step2: Calculate the total hydrogen atoms
To find the total number of hydrogen atoms, multiply the coefficient of the molecule by the number of hydrogen atoms in one molecule. So, \(2\times2 = 4\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
4