QUESTION IMAGE
Question
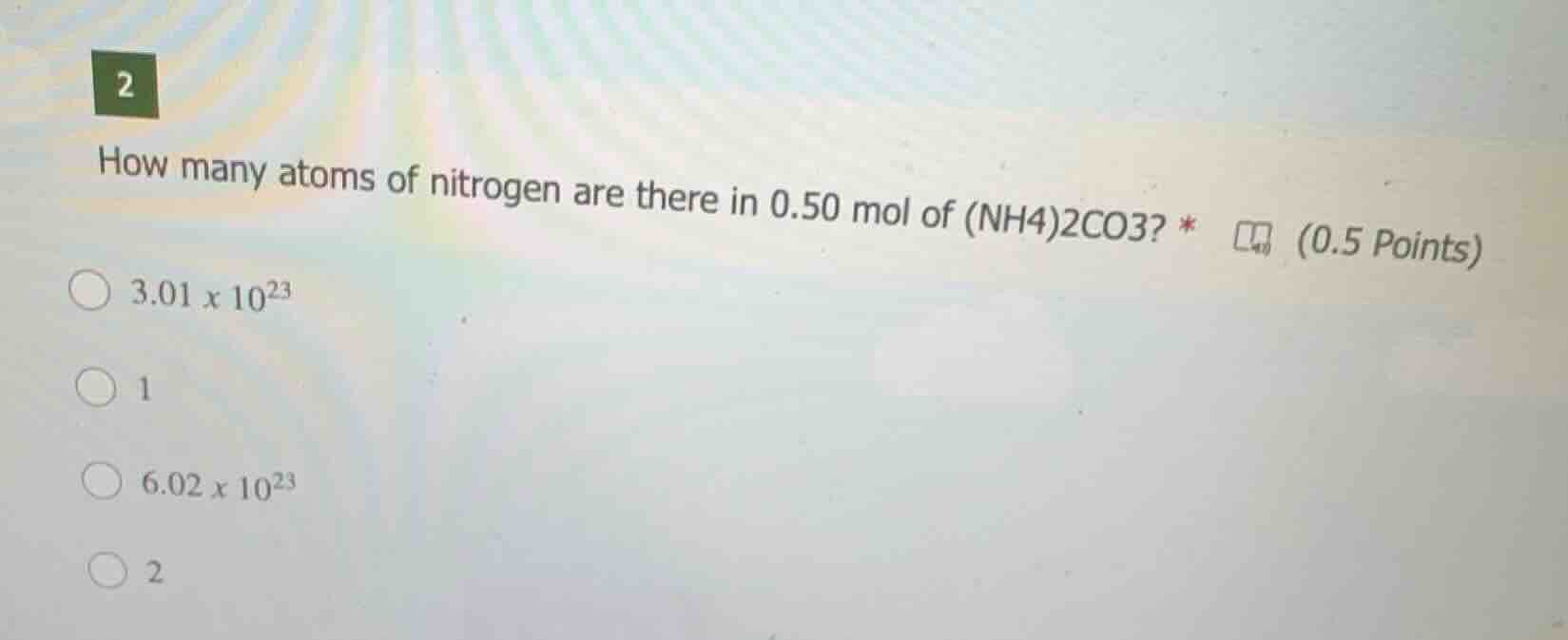
2
how many atoms of nitrogen are there in 0.50 mol of (nh4)2co3? *
3.01 x 10^23
1
6.02 x 10^23
2
(0.5 points)
Step1: Determine moles of N per mole of compound
In \((NH_4)_2CO_3\), the subscript of \(NH_4\) is 2, so each mole of \((NH_4)_2CO_3\) has 2 moles of N.
Step2: Calculate moles of N in 0.50 mol of compound
Moles of N = \(0.50\space mol\space (NH_4)_2CO_3\times\frac{2\space mol\space N}{1\space mol\space (NH_4)_2CO_3}=1.0\space mol\space N\)
Step3: Use Avogadro's number to find atoms
Avogadro's number is \(6.02\times10^{23}\space atoms/mol\). Atoms of N = \(1.0\space mol\times6.02\times10^{23}\space atoms/mol = 6.02\times10^{23}\space atoms\)? Wait, no—wait, 0.50 mol of compound, 2 N per compound: 0.50 2 = 1.0 mol N? Wait, no, wait: 0.50 mol of \((NH_4)_2CO_3\), each has 2 N. So moles of N: 0.50 2 = 1.0 mol? Then atoms: 1.0 mol 6.02e23 = 6.02e23? But wait, no—wait, the formula is \((NH_4)_2CO_3\), so the number of N atoms per formula unit is 2. So moles of N = 0.50 mol compound 2 mol N / 1 mol compound = 1.0 mol N. Then number of atoms: 1.0 mol 6.02e23 atoms/mol = 6.02e23? But wait, the options: 3.01e23, 1, 6.02e23, 2. Wait, maybe I made a mistake. Wait, 0.50 mol of compound, each compound has 2 N. So moles of N: 0.50 2 = 1.0 mol? No, wait, 0.50 mol of \((NH_4)_2CO_3\), so moles of N: 0.50 2 = 1.0 mol? Then atoms: 1.0 6.02e23 = 6.02e23. But let's check again. Wait, Avogadro's number is \(6.02\times10^{23}\) particles per mole. So if we have 0.50 mol of the compound, and each compound has 2 N atoms, then moles of N: 0.50 2 = 1.0 mol. Then number of atoms: 1.0 mol \(6.02\times10^{23}\) atoms/mol = \(6.02\times10^{23}\) atoms. Wait, but let's check the options. The third option is \(6.02\times10^{23}\). Wait, but maybe I messed up. Wait, no—wait, 0.50 mol of compound, 2 N per compound: 0.50 2 = 1.0 mol N. Then atoms: 1.0 6.02e23 = 6.02e23. So the correct option is \(6.02\times10^{23}\).
Wait, but wait, maybe I miscalculated. Let's re-express:
1 mole of \((NH_4)_2CO_3\) contains 2 moles of N (because of the 2 in front of \(NH_4\)). So 0.50 moles of \((NH_4)_2CO_3\) will contain 0.50 * 2 = 1.0 moles of N.
Number of atoms = moles Avogadro's number = 1.0 mol \(6.02\times10^{23}\) atoms/mol = \(6.02\times10^{23}\) atoms. So the answer is the third option.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(6.02\times 10^{23}\) (the option with \(6.02\times 10^{23}\))