QUESTION IMAGE
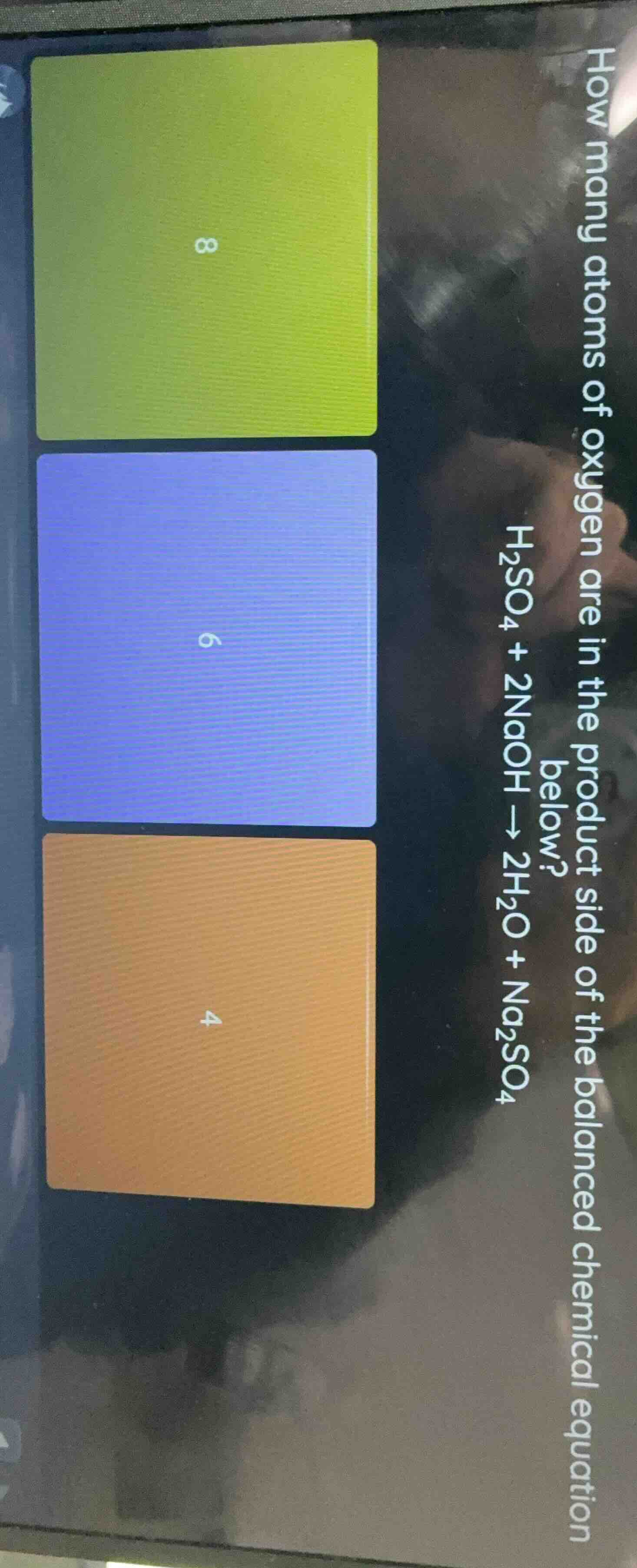
Question
how many atoms of oxygen are in the product side of the balanced chemical equation below?
h₂so₄ + 2naoh → 2h₂o + na₂so₄
8
6
4
Step1: Identify products and their oxygen atoms
The product side of the balanced chemical equation is \(2H_2O + Na_2SO_4\). For \(H_2O\): each \(H_2O\) has 1 oxygen atom, and there are 2 moles of \(H_2O\), so oxygen atoms from \(H_2O\) are \(2\times1 = 2\). For \(Na_2SO_4\): each \(Na_2SO_4\) has 4 oxygen atoms, and there is 1 mole of \(Na_2SO_4\), so oxygen atoms from \(Na_2SO_4\) are \(1\times4 = 4\).
Step2: Sum the oxygen atoms
Total oxygen atoms = oxygen from \(H_2O\) + oxygen from \(Na_2SO_4\) = \(2 + 4 = 6\). Wait, no, wait: Wait, the formula is \(2H_2O\) (so 2 molecules of \(H_2O\), each with 1 O, so 2*1=2 O) and \(Na_2SO_4\) (1 molecule, 4 O). Wait, but wait, let's re - check the equation. The balanced equation is \(H_2SO_4+2NaOH
ightarrow2H_2O + Na_2SO_4\). So for \(H_2O\): coefficient is 2, so number of O atoms in \(H_2O\) is \(2\times1 = 2\). For \(Na_2SO_4\): coefficient is 1, number of O atoms is \(1\times4 = 4\). Total O atoms: \(2 + 4=6\)? Wait, no, wait, maybe I made a mistake. Wait, \(H_2O\) has 1 O per molecule, 2 molecules: 2 O. \(Na_2SO_4\) has 4 O per molecule, 1 molecule: 4 O. Total: 2 + 4 = 6. But wait, the options are 8, 6, 4. Wait, maybe I misread the equation. Wait, the equation is \(H_2SO_4 + 2NaOH
ightarrow2H_2O+Na_2SO_4\). Let's count O atoms on product side: \(2H_2O\): 2*1 = 2 O. \(Na_2SO_4\): 4 O. Total: 2 + 4 = 6. So the answer should be 6.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6