QUESTION IMAGE
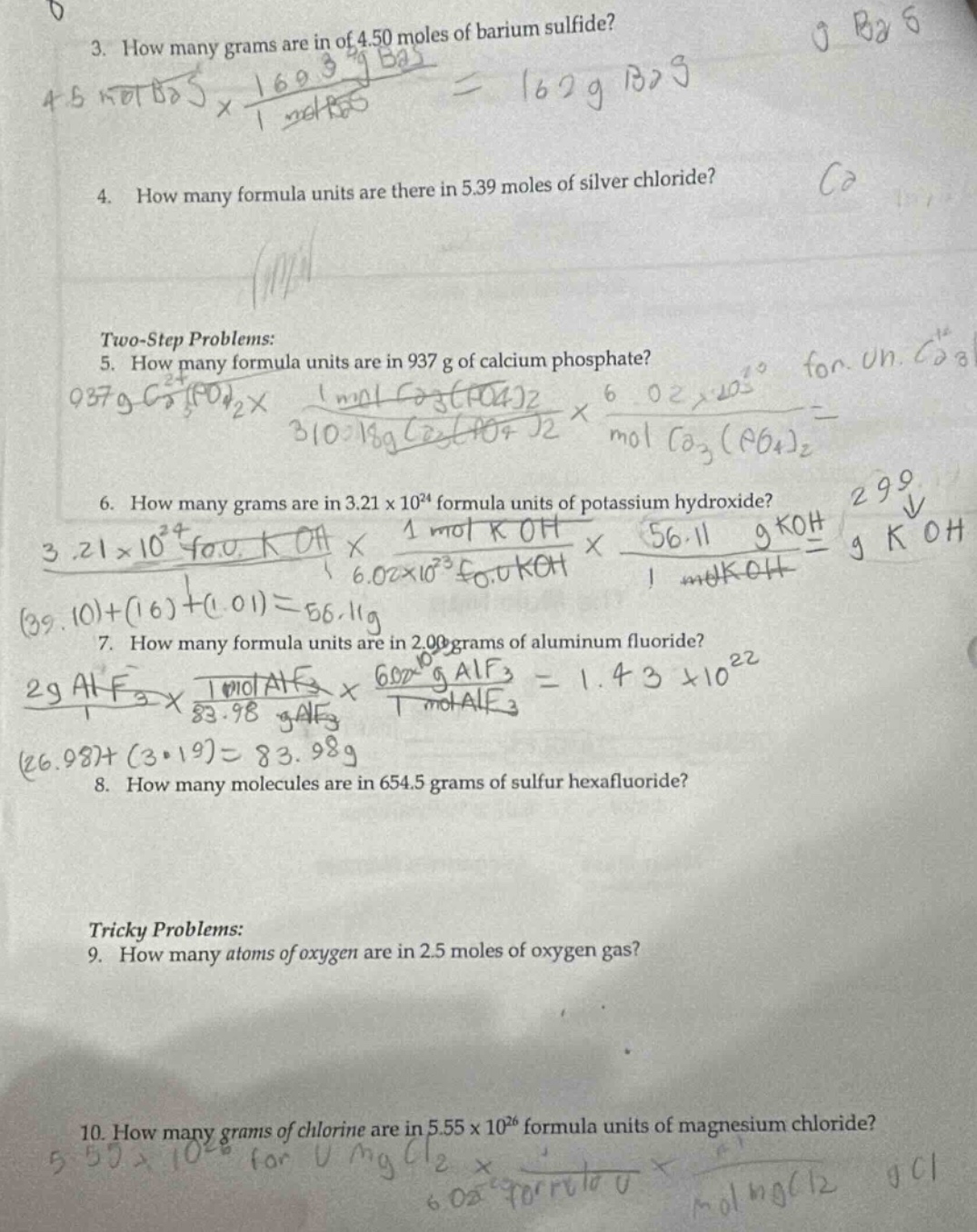
Question
- how many grams are in of 4.50 moles of barium sulfide?
- how many formula units are there in 5.39 moles of silver chloride?
two - step problems:
- how many formula units are in 937 g of calcium phosphate?
- how many grams are in 3.21×10²⁴ formula units of potassium hydroxide?
- how many formula units are in 2.00 grams of aluminum fluoride?
- how many molecules are in 654.5 grams of sulfur hexafluoride?
tricky problems:
- how many atoms of oxygen are in 2.5 moles of oxygen gas?
- how many grams of chlorine are in 5.55×10²⁶ formula units of magnesium chloride?
Problem 3: How many grams are in 4.50 moles of barium sulfide?
Step 1: Determine the molar mass of BaS
Barium (Ba) has a molar mass of \( 137.33 \, \text{g/mol} \), sulfur (S) has a molar mass of \( 32.07 \, \text{g/mol} \). So, molar mass of \( \text{BaS} = 137.33 + 32.07 = 169.40 \, \text{g/mol} \).
Step 2: Calculate mass from moles
Use the formula \( \text{mass} = \text{moles} \times \text{molar mass} \). Substitute moles \( = 4.50 \, \text{mol} \) and molar mass \( = 169.40 \, \text{g/mol} \).
\( \text{mass} = 4.50 \, \text{mol} \times 169.40 \, \text{g/mol} \)
Step 1: Recall Avogadro's number
Avogadro's number is \( 6.022 \times 10^{23} \, \text{formula units/mol} \).
Step 2: Calculate formula units
Use the formula \( \text{formula units} = \text{moles} \times \text{Avogadro's number} \). Substitute moles \( = 5.39 \, \text{mol} \).
\( \text{formula units} = 5.39 \, \text{mol} \times 6.022 \times 10^{23} \, \text{formula units/mol} \)
Step 1: Recall Avogadro's number
Avogadro's number is \( 6.022 \times 10^{23} \, \text{formula units/mol} \).
Step 2: Calculate formula units
Use the formula \( \text{formula units} = \text{moles} \times \text{Avogadro's number} \). Substitute moles \( = 5.39 \, \text{mol} \).
\( \text{formula units} = 5.39 \, \text{mol} \times 6.022 \times 10^{23} \, \text{formula units/mol} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 762.3 \, \text{grams} \) (or \( 762 \, \text{g} \) if rounded to three significant figures)