QUESTION IMAGE
Question
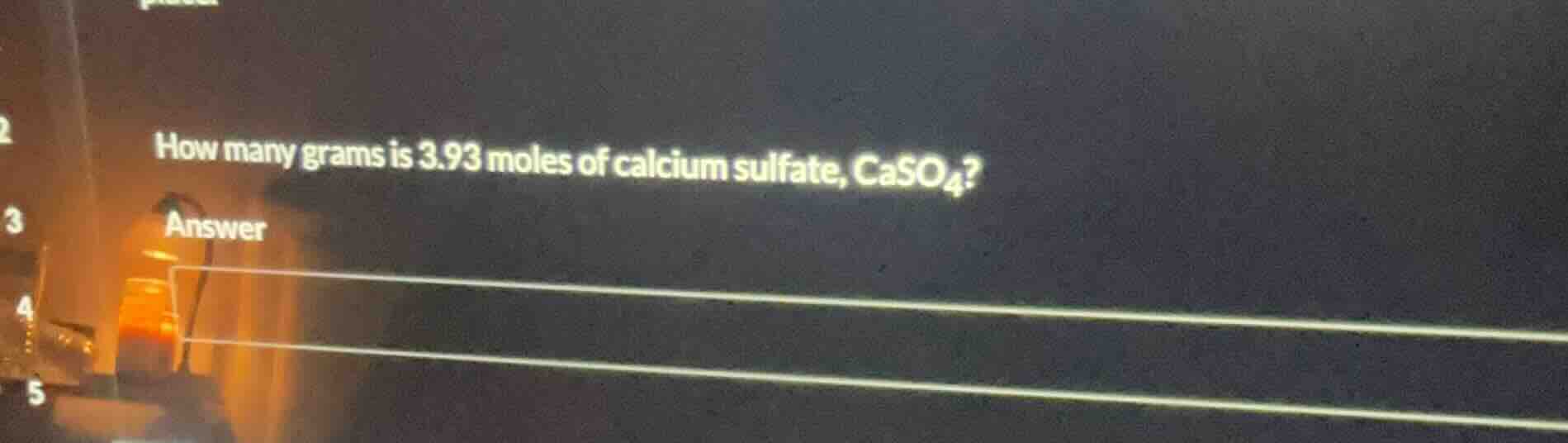
how many grams is 3.93 moles of calcium sulfate, caso₄? answer
Step1: Calculate molar mass of \( \text{CaSO}_4 \)
Molar mass of \( \text{Ca} = 40.08 \, \text{g/mol} \), \( \text{S} = 32.07 \, \text{g/mol} \), \( \text{O} = 16.00 \, \text{g/mol} \).
Molar mass of \( \text{CaSO}_4 = 40.08 + 32.07 + (4 \times 16.00) = 40.08 + 32.07 + 64.00 = 136.15 \, \text{g/mol} \).
Step2: Use moles to grams formula (\( \text{mass} = \text{moles} \times \text{molar mass} \))
Given moles \( = 3.93 \, \text{mol} \), molar mass \( = 136.15 \, \text{g/mol} \).
Mass \( = 3.93 \, \text{mol} \times 136.15 \, \text{g/mol} \approx 535 \, \text{g} \) (rounded to three significant figures).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \approx 535 \) grams