QUESTION IMAGE
Question
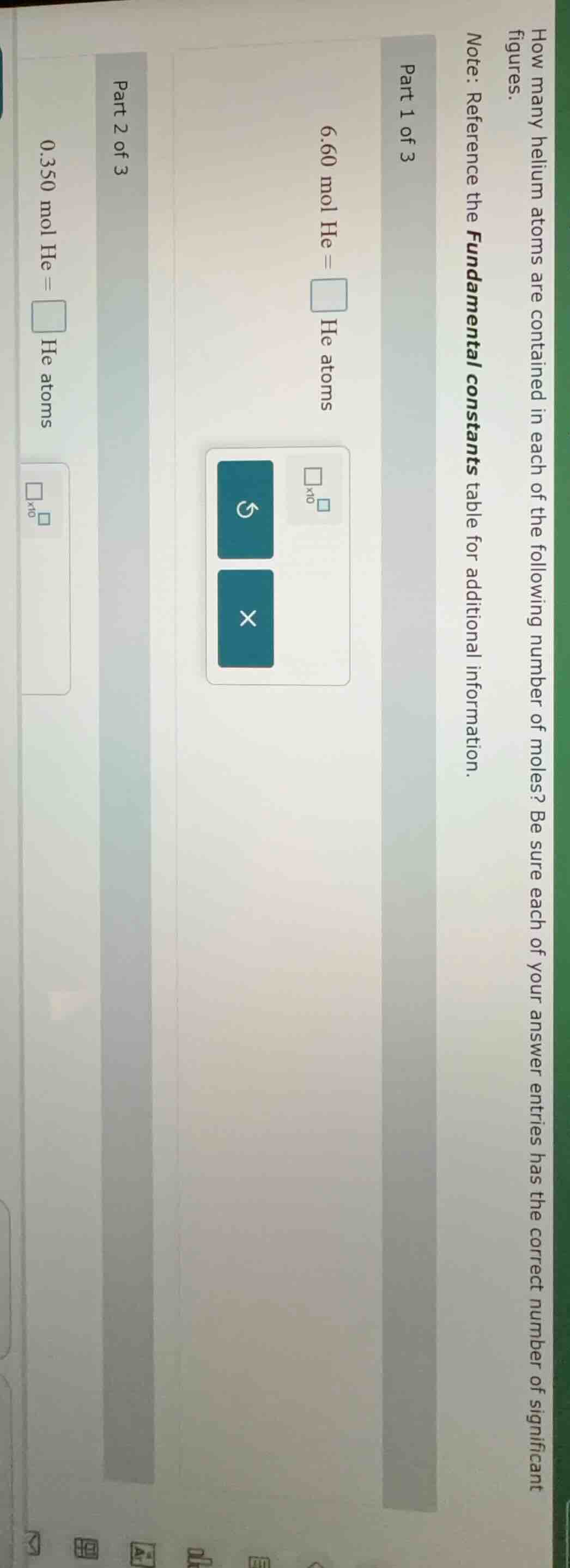
how many helium atoms are contained in each of the following number of moles? be sure each of your answer entries has the correct number of significant figures.
note: reference the fundamental constants table for additional information.
part 1 of 3
6.60 mol he = \square he atoms
part 2 of 3
0.350 mol he = \square he atoms
Part 1 of 3
Step 1: Recall Avogadro's number
Avogadro's number is \( 6.022 \times 10^{23} \) atoms/mol. To find the number of atoms, we multiply the number of moles by Avogadro's number.
Step 2: Calculate the number of He atoms
Given \( 6.60 \) mol He, the number of atoms \( N = n \times N_A \), where \( n = 6.60 \) mol and \( N_A = 6.022 \times 10^{23} \) atoms/mol.
\[
N = 6.60 \, \text{mol} \times 6.022 \times 10^{23} \, \text{atoms/mol}
\]
\[
N = 6.60 \times 6.022 \times 10^{23}
\]
\[
N \approx 3.97452 \times 10^{24}
\]
Rounding to three significant figures (since 6.60 has three), we get \( 3.97 \times 10^{24} \) (or using the calculator's precision, maybe \( 3.97 \times 10^{24} \) or more accurately \( 3.97452\times 10^{24}\approx 3.97\times 10^{24}\) when considering significant figures). But let's do the exact multiplication: \( 6.60\times6.022 = 39.7452\), so \( 39.7452\times 10^{23}=3.97452\times 10^{24}\approx 3.97\times 10^{24}\) (or if we use \( 6.02\times 10^{23}\) for simplicity, \( 6.60\times6.02\times 10^{23}= 39.732\times 10^{23}=3.9732\times 10^{24}\approx 3.97\times 10^{24}\)).
Wait, actually, let's check the significant figures. 6.60 has three, Avogadro's number is often taken as \( 6.022\times 10^{23} \) (four significant figures), so the result should have three. So \( 6.60\times6.022\times 10^{23}= 39.7452\times 10^{23}=3.97452\times 10^{24}\approx 3.97\times 10^{24}\) (or \( 3.97\times 10^{24} \) when rounded to three significant figures). But maybe the system expects using \( 6.02\times 10^{23} \), let's recalculate: \( 6.60\times6.02 = 39.732 \), so \( 39.732\times 10^{23}=3.9732\times 10^{24}\approx 3.97\times 10^{24}\).
Alternatively, using a calculator: \( 6.60 \times 6.02214076\times 10^{23} = 6.60\times6.02214076 = 39.746129016 \), so \( 39.746129016\times 10^{23}=3.9746129016\times 10^{24}\approx 3.97\times 10^{24}\) (three significant figures).
Step 1: Recall Avogadro's number
Avogadro's number is \( 6.022 \times 10^{23} \) atoms/mol. To find the number of atoms, we multiply the number of moles by Avogadro's number.
Step 2: Calculate the number of He atoms
Given \( 0.350 \) mol He, the number of atoms \( N = n \times N_A \), where \( n = 0.350 \) mol and \( N_A = 6.022 \times 10^{23} \) atoms/mol.
\[
N = 0.350 \, \text{mol} \times 6.022 \times 10^{23} \, \text{atoms/mol}
\]
\[
N = 0.350 \times 6.022 \times 10^{23}
\]
\[
N \approx 2.1077 \times 10^{23}
\]
Rounding to three significant figures (since 0.350 has three), we get \( 2.11 \times 10^{23} \) (or more accurately, \( 2.1077\times 10^{23}\approx 2.11\times 10^{23}\) with three significant figures).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 3.97 \times 10^{24} \) (or in the box, if we consider the format, maybe \( 3.97\times10^{24} \), but let's check the exact calculation. Wait, maybe the problem expects using \( 6.02\times 10^{23} \), so \( 6.60\times6.02\times 10^{23}= 39.732\times 10^{23}=3.9732\times 10^{24}\approx 3.97\times 10^{24}\). So the answer is \( 3.97 \times 10^{24} \) (or \( 3.97\times10^{24} \)).