QUESTION IMAGE
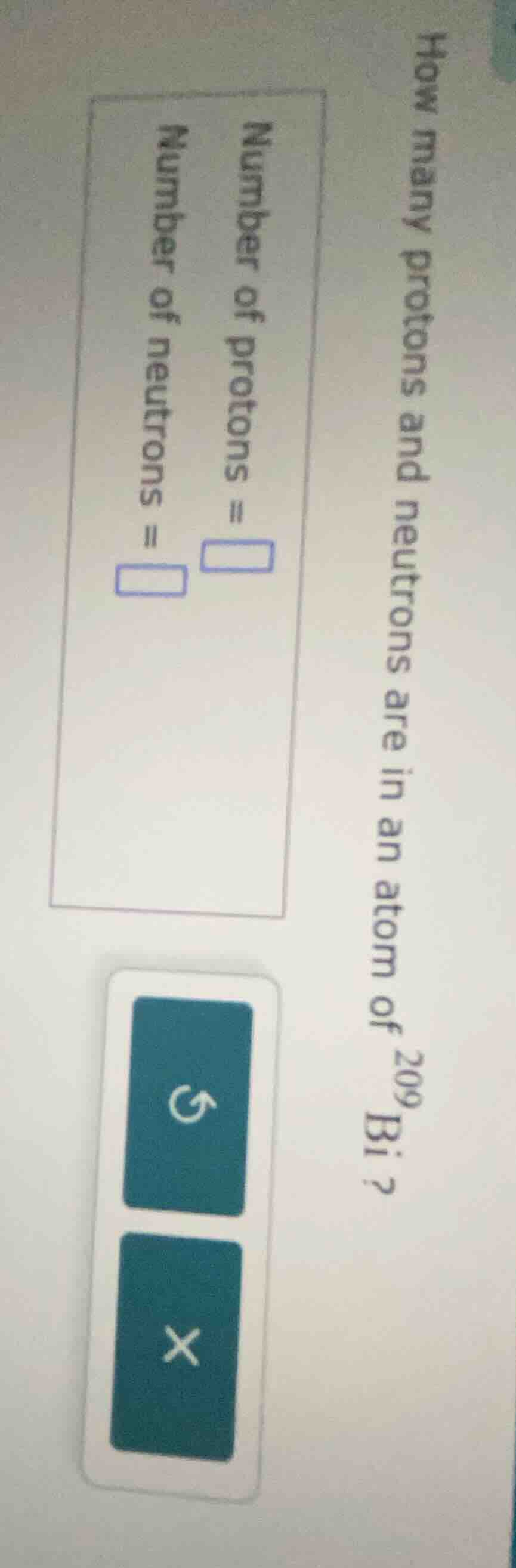
Question
how many protons and neutrons are in an atom of $^{209}$bi?
number of protons =
number of neutrons =
Step1: Find atomic number of Bi
Bismuth (Bi) has an atomic number of 83, so number of protons = atomic number = 83.
Step2: Calculate number of neutrons
Mass number (209) = number of protons + number of neutrons. So number of neutrons = mass number - number of protons = 209 - 83 = 126.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Number of protons = 83, Number of neutrons = 126